��Ŀ����
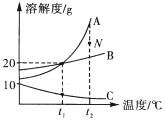
����Ŀ����ˮƿ�þ���ƿ���ڱ�����������ˮ������ɷ���̼���ơ�������þ������ˮ��ʵ��������һƿˮ����Ʒ��Ϊ�����������ɷݵ�����������ij��ѧ��ȤС����ͬѧ������ͼ��ʾװ������ʵ�飨��װ�����������ã�A��C��D����װҩƷ����������֪����ʯ�������������������ƵĻ������������£�������þ�ֽ⣬��Ӧ�Ļ�ѧ����ʽΪ��Mg(OH)2![]() MgO+H2O����
MgO+H2O����
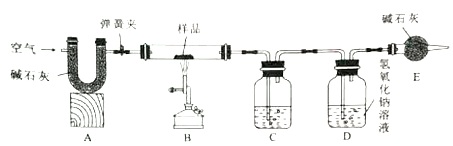
����ʵ�鲽�����£�
I.������Ϊm����Ʒװ��װ��B���������У���ͼ���Ӻ�װ�����رյ���������Ʒ���ȣ�
II������Ʒ��ȫ��Ӧ����������ͨ���������Ϩ���ƾ������ֱ����������ȴ��
III.ʵ����ϣ�����װ��C��D�е�Һ�����Էֱ�������m1��m2��
IV.����ʵ���������ݼ������Ʒ��̼���ơ�������þ��ˮ������������
�������������⣺
��1��װ��C����װҩƷΪ___________��
��2��װ��D�з�����Ӧ�Ļ�ѧ����ʽΪ___________��
��3����ˮ����Ʒ��̼��Ƶ����������ı���ʽΪ___________��
��4��ʵ�鷴˼��ʵ�鲽�費������û��װ��A���ᵼ��������þ���ⶨ���_____������ƫ������ƫС���������ж���)��
���𰸡���1��Ũ���ᣨ2��CO2+2NaOH=Na2CO3+H2O��3��![]() ������������
������������
��4��ƫС
������������������Ϣ֪����1��װ��C����װҩƷΪŨ���ᡣ��2��װ��D�з�����Ӧ�Ļ�ѧ����ʽΪCO2+2NaOH=Na2CO3+H2O����3����ˮ����Ʒ��̼��Ƶ����������ı���ʽΪ![]() ��100������4��ʵ�鷴˼��ʵ�鲽�費�䣬��û��װ��A���ᵼ��������þ�IJⶨ���ƫС��
��100������4��ʵ�鷴˼��ʵ�鲽�費�䣬��û��װ��A���ᵼ��������þ�IJⶨ���ƫС��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�