��Ŀ����
����Ŀ����ˮƿ�þú�ƿ���ڱڳ�����һ��ˮ������ɷ���̼��ơ�������þ������ˮ��ʵ��������һƿˮ����Ʒ��Ϊ�ⶨ���и��ɷݵ�����������ij��ѧ��ȤС���ͬѧ������ͼ��ʾװ�ý���ʵ�顣
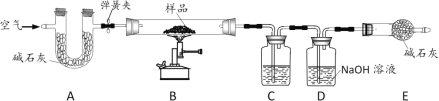
��֪��
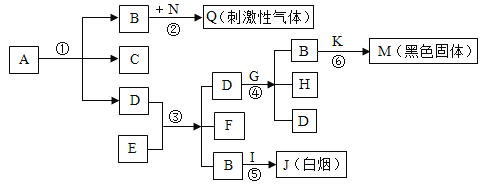
��װ����������ã�A��C��D����װҩƷ��Ϊ������
�ڼ�ʯ���������ƺ��������ƵĹ������
���ڼ��������£�������þ�ֽ�����������
��Ҫʵ�鲽�����£�
I��������Ϊ12.0g����Ʒװ��װ��B�IJ������У�����װ�ã��رյ��ɼУ����ȣ�
II������Ʒ��ȫ��Ӧ���� ____��ѡ�������ɼл���ͨ�����������Ϩ��ƾ����������
III��ʵ����ϣ����װ��C��D�������ֱ�������2.5g��3.3g��
IV������ʵ���������ݼ������Ʒ��̼��ơ�������þ��ˮ������������
��ش��������⣺
��1��װ��C����װҩƷΪ_____��
��2��װ��D�з�����Ӧ�Ļ�ѧ����ʽΪ_____��
��3����ˮ����Ʒ��̼��Ƶ���������Ϊ_____��
��4��װ��E������Ϊ_____��
��5��ʵ�鷴˼��ʵ�鲽�費�䣬��û��װ��A���ᵼ��̼��ƵIJⶨ���_____������ƫ��������ƫС���������ж�������ͬ) ����û��װ��E���ᵼ��������þ�IJⶨ��� ____��
���𰸡��ȴ��ɼл���ͨ����� Ũ���ᣨŨH2SO4�� 2NaOH+CO2=Na2CO3+H2O 62.5% �����������еĶ�����̼��ˮ����ֹ����Dװ�� ƫ�� ƫС
��������
ʵ�鲽��II��Ϊ��ֹC��Һ�嵹������������������C��D�����գ�����Ʒ��ȫ��Ӧ���ȴ��ɼл���ͨ�������Ϩ��ƾ���ƣ�
��1�����ݡ�ƿ���ڱڳ�����һ��ˮ������ɷ���̼��ơ�������þ������ˮ���͡��ڼ��������£�������þ�ֽ�������������֪��ˮ�����Ⱥ�����������д���ˮ���е�ˮ��������þ�ֽ����ɵ�ˮ��Ũ���������ˮ�ԣ�װ��C����װҩƷΪŨ���ᣬ������������ˮ�����Ⱥ������ˮ������
��2��װ��D���������ƺͷ�Ӧ���ɵĶ�����̼��Ӧ����̼���ƺ�ˮ��������Ӧ�Ļ�ѧ����ʽΪ��2NaOH+CO2�TNa2CO3+H2O��
��3�����������֪��D�������ֱ�������3.3g��̼��Ʒֽ�����Ķ�����̼������
�裺̼�������Ϊx��

![]() x=7.5g��
x=7.5g��
��ˮ����Ʒ��̼��Ƶ���������=![]()
��4��װ��E������Ϊ�����������еĶ�����̼��ˮ����ֹ����Dװ�ã�
��5��ʵ�鲽�費�䣬��û��װ��A�������еĶ�����̼�ܱ�����������Һ���գ��ᵼ��̼��Ƶ���������ƫ����û��װ��E�������еĶ�����̼�ܱ�����������Һ���գ��ᵼ��̼��Ƶ���������ƫ��Ӷ�����������þ�IJⶨ���ƫС��