��Ŀ����
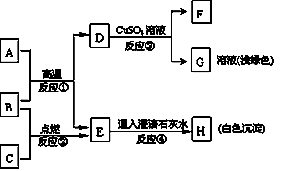
��ѧϰ�����У�С��ͬѧ���һ�����⣺��̼ȼ��ʱ��������CO2��Ҳ��������C0����ô̼������ͭ��Ӧ���ɵ�����Ҳ�ж��ֿ���?��
[����]̼������ͭ��Ӧ���ɵ������У���ֻ��CO2 ��ֻ��CO�ۼ�CO2������CO��
[��������]������������Һ���Ȼ�����ֽ��CO2�ޱ仯����������CO�����������ɫ��
[�������]ͨ���������Ϻ�����֪ʶ��С����������ý�����������Һ���Ȼ�����ֽ�ͳ����ʯ��ˮ��̼������ͭ��Ӧ���ɵ�������м��顣����С������룬���������գ�
(1)����ֽ����ɫ��ʯ��ˮ����ǣ���ֻ��C02��
(2)����ֽ ��ʯ��ˮ ����ֻ��C0��
(3)����ֽ ��ʯ��ˮ �������CO2������CO��
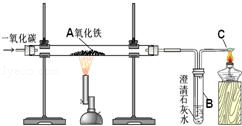
[ʵ����֤]����˼��֮��С���������ͼ��ʾʵ��װ�ã�
ʵ�鲽�裺
(1)���ɼУ���ͨһ��������������N2��
(2)�رյ��ɼУ���ȼ�ƾ��ƣ����ȡ�
���ۣ�С��ͨ������ʵ������ȷ�ϲ��������ȷ�ģ�����д��װ��C�з�����Ӧ�Ļ�ѧ����ʽ�� ��
[��˼������]
(1)��ʵ�鿪ʼͨһ���N2��Ŀ���� ��
(2)��ӻ����ĽǶȷ�����ʵ��װ�õIJ�����֮�� ��
[�������](2)��������������
(3)���� �����
[ʵ����֤] Ca(OH)2 + CO2 =CaCO3��+ H2O
[��˼������](1)�ų�װ���ڵĿ��� (2)û��β������(����)װ�á�
����