��Ŀ����
������л�ѧ����ʽ������Ҫ��ش����⣮
��1����˿��������ȼ�գ�
��2���õ����ɫ�����ĸ��ֽⷴӦ��
��3������狀��������Ʒ�Ӧ��ͬʱ����һ�ְ���ζ�����壺
��1����˿��������ȼ�գ�
3Fe+2O2
Fe3O4
| ||
3Fe+2O2
Fe3O4
��������Ӧ������
| ||
���Ϸ�Ӧ
���Ϸ�Ӧ
����2���õ����ɫ�����ĸ��ֽⷴӦ��
FeCl3+3NaOH�TFe��OH��3��+3NaCl
FeCl3+3NaOH�TFe��OH��3��+3NaCl
����3������狀��������Ʒ�Ӧ��ͬʱ����һ�ְ���ζ�����壺
��NH4��2SO4+2NaOH�TNa2SO4+2NH3��+2H2O
��NH4��2SO4+2NaOH�TNa2SO4+2NH3��+2H2O
������������ʪ�����ɫʯ��
��ɫʯ��
��ֽ����������д��ѧ����ʽʱ��������Ϥ��Ӧ�������ͷ�Ӧ�������������ݿ���ʵ�����������غ㶨�ɣ����Ծݴ���ɸ����л�ѧ����ʽ����д��
����⣺��1����˿��������ȼ��������������������ѧ����ʽΪ��3Fe+2O2
Fe3O4���÷�Ӧ�Ƕ������ʷ�Ӧ����һ�����ʣ������ڻ��Ϸ�Ӧ��
��2���Ȼ������������Ʒ�Ӧ���ɺ��ɫ�����������������Ȼ��ƣ��䷴Ӧ�Ļ�ѧ����ʽ��Ϊ��FeCl3+3NaOH�TFe��OH��3��+3NaCl��
��3������狀��������Ʒ�Ӧ���������ơ�ˮ�Ͱ���������Ϊ�������壬��ʹʪ��ĺ�ɫʯ����ֽ���������Կ����ú�ɫʯ����ֽ��֤��
�ʴ�Ϊ����1��3Fe+2O2
Fe3O4�����Ϸ�Ӧ��
��2��FeCl3+3NaOH�TFe��OH��3��+3NaCl��
��3����NH4��2SO4+2NaOH�TNa2SO4+2NH3��+2H2O����ɫʯ�
| ||
��2���Ȼ������������Ʒ�Ӧ���ɺ��ɫ�����������������Ȼ��ƣ��䷴Ӧ�Ļ�ѧ����ʽ��Ϊ��FeCl3+3NaOH�TFe��OH��3��+3NaCl��
��3������狀��������Ʒ�Ӧ���������ơ�ˮ�Ͱ���������Ϊ�������壬��ʹʪ��ĺ�ɫʯ����ֽ���������Կ����ú�ɫʯ����ֽ��֤��
�ʴ�Ϊ����1��3Fe+2O2
| ||
��2��FeCl3+3NaOH�TFe��OH��3��+3NaCl��
��3����NH4��2SO4+2NaOH�TNa2SO4+2NH3��+2H2O����ɫʯ�
������������Ҫ�����˻�ѧ����ʽ����ȷ��д�ķ������裬�˽����ʷ�����ѧ�仯ʱ�������仯�����շ�Ӧ���͵��ж�������

��ϰ��ϵ�д�
�����Ŀ
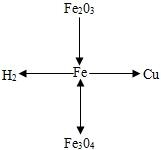 ��ѧ��ͨ���á�������ʾ���������ͷ��ָʾ����ת�����á���������ʾ��ͷ��������֮����һ�������¿����ת�����Ӷ�����������֮��Ĺ�ϵ�ü�����ͼ��ʾ��������ʾ������
��ѧ��ͨ���á�������ʾ���������ͷ��ָʾ����ת�����á���������ʾ��ͷ��������֮����һ�������¿����ת�����Ӷ�����������֮��Ĺ�ϵ�ü�����ͼ��ʾ��������ʾ������