��Ŀ����
����Ŀ��������Դ����Լ��Դ���������������Ǵ�ҹ�ͬ��ע�����⣮
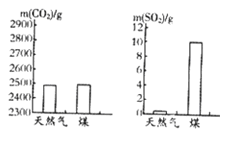
��1�����ȼ��1000g��Ȼ����ú��������CO2��SO2�����������ͼ��ʾ���� ȼ�նԻ���Ӱ���С��1000gú����Ԫ�ص�����Ϊ ��������Ԫ����ȫת��ΪSO2����
��2��SO2�Ĵ����ŷ����γ����꣬pH 5.6����д����������������=�����Ľ����Ϊ���꣬��úȼ�պ�����ͨ���������������ܡ�ʯ��ˮ���С����ɼ���SO2���ŷţ�ʯ��ˮ��Ҫ�����ܡ���Ŀ���� ��ʯ��ˮ��SO2��Ӧ�Ļ�ѧ����ʽΪ ��SO2�ͼӦ��CO2���ƣ���
��3��Ŀǰ��������β��ϵͳ�а�װ��ת�����ɽ���Ⱦ��CO��NOת��Ϊ����CO2��N2��2CO+2NO![]() 2CO2+N2������ݡ��ڻ�ѧ��Ӧ��������Ԫ�ػ��ϼ۱仯�Ļ�ѧ��Ӧ������������ԭ��Ӧ���жϣ��÷�Ӧ ����д���ǡ����ǡ���������ԭ��Ӧ��
2CO2+N2������ݡ��ڻ�ѧ��Ӧ��������Ԫ�ػ��ϼ۱仯�Ļ�ѧ��Ӧ������������ԭ��Ӧ���жϣ��÷�Ӧ ����д���ǡ����ǡ���������ԭ��Ӧ��
���𰸡���1��5g��2�������������������գ�SO2 +Ca��OH��2 �TCaSO3+H2O��3����
��������
�����������1�����ͼʾ��֪����Ȼ�������Ķ���������١���������ȼ��ȼ�նԻ���Ӱ���С������Ȼ����
��ͼ��֪1000gú����10g�����������ݶ�������Ļ�ѧʽ��֪��������Ԫ�ص�����Ϊ10g��![]() ��100%=5g��
��100%=5g��
��2��������ָpH��5.6����ˮ�������ܡ�ʯ��ˮ�����ڶ����������������ƵĽӴ���ʹ���ַ�Ӧ������������֪��SO2��ʯ��ˮ��������Ӧ��������ƺ�ˮ��������Ӧ�Ļ�ѧ����ʽΪSO2 +Ca��OH��2 �TCaSO3+H2O��
��3�����������֪������Ԫ�ػ��ϼ۱仯�Ļ�ѧ��Ӧ�ͽ���������ԭ��Ӧ����Ӧǰ��Ԫ����̼Ԫ���Ļ��ϼ��������˸ı䣬�����÷�Ӧ����������ԭ��Ӧ��