��Ŀ����
����Ŀ����ʵġ�̼�������˵����������һ���߽���̼�������硣
��1�������±��ṩ����Ϣ����д�йغ�̼���ʵĶ�Ӧ���ʡ�
������; | ���ʯ�и�� | ʯī���缫 | ����̿��ˮ |
��Ӧ���� | ��________ | ��________ | ��________ |
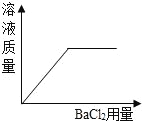
��2��Һ̬������̼������������˾ȵ��������ҷ����Ļ��֣�����˵����ȷ����_____�����ţ�

A.Һ̬������̼��������Ⱦ��������
B.������̼�ɸ�����ȼ������棬����������
C.Һ̬������̼����ʱ���ȣ������˿�ȼ����Ż�㡣
��3������Ķ�����̼�Ӿ��ˡ�����ЧӦ����д��һ�����ٶ�����̼�ŷŵĽ���________��
��4��������̼��һ�ֱ������Դ���̶������ö�����̼��һ���ɹ������ǣ��ڸ��¸�ѹ�£�������̼�Ͱ�����NH3�����Ժϳ�����CO(NH2)2,ͬʱ����ˮ���÷�Ӧ�Ļ�ѧ����ʽΪ________��
��5��Ŀǰ�������Ի�ʯȼ��Ϊ��Ҫ��Դ����ʯȼ����ú��________����Ȼ�������Ƕ�����________�������������������������Դ��
��6����Ȼ���м�����ȫȼ�յĻ�ѧ����ʽΪ________��
��7����ú��ȣ�����Ȼ����ȼ�ϵ��ŵ���________��
���𰸡�Ӳ�ȴ����ԣ�������ABֲ�����֣����������𰸣�CO2+2NH3 ![]() CO(NH2)2+H2Oʯ�ͣ���������CH4+2O2
CO(NH2)2+H2Oʯ�ͣ���������CH4+2O2 ![]() CO2+2H2O��ֵ�ߣ������Ķ�����̼�٣��Ի�����Ӱ��С
CO2+2H2O��ֵ�ߣ������Ķ�����̼�٣��Ի�����Ӱ��С
��������
���⿼����̼���ʵ��������ʼ���;��������̼����;��������̼�Ի�����Ӱ������ѧ����ʽ��д�����ѶȲ������������е�֪ʶ���з������
��1�����ʯ��Ӳ������������и����ʯī���е����ԣ����������缫������̿���������ԣ��������ھ�ˮ�����
������; | ���ʯ�и�� | ʯī���缫 | ����̿��ˮ |
��Ӧ���� | ��Ӳ����� | �ڵ����� | �������� |
��2��A��Һ̬������̼��������Ⱦ�������ϣ���A��ȷ��B��������̼���ܶȱȿ������ܶȴɸ�����ȼ������棬�����������ﵽ����Ŀ�ģ���B��ȷ��C��Һ̬������̼����ʱ���ȣ������¶�ʹ��ﵽ��ȼ����Ż�����£������ܽ������ʵ��Ż�㣬��C����ѡAB��
��3�����ٶ�����̼�ŷŵĴ�ʩ�м��ٻ�ʯȼ�ϵ�ʹ�á���ʹ��������Դ��ֲ�����ֵȣ����ֲ�����֣���ʹ�������Դ�ȣ���
��4���ڸ��¸�ѹ�£�������̼�Ͱ�����NH3�����Ժϳ�����CO(NH2)2,ͬʱ����ˮ���÷�Ӧ�Ļ�ѧ����ʽΪCO2+2NH3 ![]() CO(NH2)2+H2O��
CO(NH2)2+H2O��
��5��Ŀǰ�������Ի�ʯȼ��Ϊ��Ҫ��Դ����ʯȼ����ú��ʯ�ͺ���Ȼ�������Ƕ�����
����������Դ��
��6��������ȫȼ�ն�����̼��ˮ���÷�Ӧ�Ļ�ѧ����ʽΪCH4+2O2 ![]() CO2+2H2O��
CO2+2H2O��
��7����Ȼ����һ�ֽྻ������������Դ�����������۳��������к����ʣ�ȼ��ʱ����������̼����ú���������ЧӦ�ϵͣ��ܴӸ����ϸ��ƻ�����������Ȼ������ֵ�ߣ���ú�������Ȼ����ȼ�ϵ��ŵ�����ֵ�ߣ������Ķ�����̼�٣��Ի�����Ӱ��С��

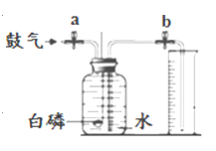
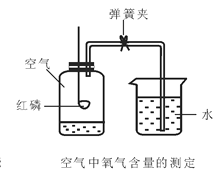
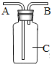
����Ŀ��������һ�ֱ������Ȼ��Դ��ij��ȤС���ͬѧ��ʵ����������ͼ��ʾװ�ô��Բⶨ�����������ĺ�����
��ʵ��عˣ�
��1��д������ȼ�յ����ֱ���ʽ��____________��
��2��ʵ��ԭ�������ں���ȼ�����Ŀ����е�������ʹƿ��____________��С���ձ���ˮ����������ƿ����װ�õ����������ã������淶������Ͳ��������ƿ��ˮ��������ܴ��Բ�ÿ����������ĺ�����
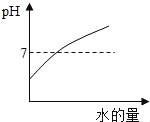
��������⣩��ʵ�����ϱ�����ȼ�չ����е����������������7% ʱ������������ȼ�գ����ͨ������ʵ�飬�ⶨ���������ֵ���ϴ���ô���ܷ����һ��ʵ��װ��ʹ�ⶨ�����Ϊȷ�أ�
���������ϣ��������ԭ���ǣ���������е�������ˮ�����ȷ�����ѧ��Ӧ������ʳ��ˮ������̿����������⡣
��ʵ��Ľ�������һ����ͬѧ�Ǹ������ڿ����������ԭ�������ͼ��ʵ��װ�ã��ٴβⶨ����������������

���ʵ���������±���
������Ŀ | ʵ��ǰ | ʵ��� | |
�ձ���ˮ����� | �ձ���ʣ��ˮ����� | ����ƿ���۳�������͵��ܵ���� | |
���/ml | 80.0 | 54.5 | 126.0 |
���������
(3)��ʵ��ԭ���Ƕȷ������Ľ����ʵ������ǰ��ȷ�ȸ��ߵ�ԭ���ǣ���_______________����____________��
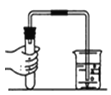
��������ͬѧ�����������ͼ��ʾ��ʵ��װ�ã�ʵ�鲽�����£�װ�����������ã����ֲ�������ȥ����
��.��ֹˮ��a��b������ƿ�л�������һ����������������������Ӵ�����ý�����Ͳ��ˮ�����ΪV1��II���ر�a��bǿ��������ȼ���ף�III������Ϩ����ȴ�����£���ֹˮ��b�����Ͳ��ˮ�������ΪV2��IV������������������������������IV�У���������������������___��