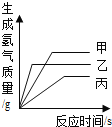
��Ŀ����
����Ŀ��ʵ���Ҳ���������װ������ͼ��ʾ����ش��������⡣
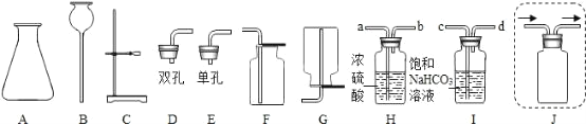
��1��ʵ���ҳ���ʯ��ʯ��ϡ������ȡ������̼���壬����װ�ÿ�ѡ��ͼ�е�_____������������ĸ��������װ����Ӧ�Ļ�ѧ����ʽΪ_____�����������̼�ķ�����_____����Ӧ�Ļ�ѧ����ʽΪ_____��
��2����������������ȡ�Ķ�����̼�г������Ȼ��������ˮ������Ϊ��ô���������Ķ�����̼���壬��ѡ����ͼ��ʾ��Hװ�ú�Iװ�ý��г��Ӻ�����ܿ����ӵ���ȷ˳���ǣ�������_____��F��
��3��Ϊ�˷�ֹ��ȡ��CO2�к�������ӷ�����HCl���壬_____����ܡ����ܡ�����û�лӷ��Ե�ϡ�������ϡ���ᣬԭ����_____��
��4��ʵ����Ҳ������������װ����ȡ����������ͼ��ʾJװ�ò��á��ſ��������ռ�����������ͼ�н����ܲ�������_____��
���𰸡�ABCD CaCO3+2HCl=CaCl2+H2O+CO2�� ������ͨ�����ʯ��ˮ�� Ca(OH)2+CO2=CaCO3![]() +H2O c��d��a��b ���� ���ɵ�����Ƹ�����ʯ��ʯ�ı��棬��ֹ��Ӧ�Ľ�һ������
+H2O c��d��a��b ���� ���ɵ�����Ƹ�����ʯ��ʯ�ı��棬��ֹ��Ӧ�Ľ�һ������ 
��������
��1��ʵ���ҳ���ʯ��ʯ��ϡ������ȡ������̼���壬���ڹ����Һ���ϲ���Ҫ���ȣ�����װ�ÿ�ѡ��ͼ�е�A��B��C��D������װ����Ӧ�Ļ�ѧ����ʽΪCaCO3+2HCl=CaCl2+H2O+CO2����������̼��ʹ����ʯ��ˮ����ǣ�ͨ�����ó���ʯ��ˮ�������Ƿ�Ϊ������̼���䷴Ӧ����ʽΪ��Ca(OH)2+CO2=CaCO3![]() +H2O�����ABCD��CaCO3+2HCl=CaCl2+H2O+CO2����������ͨ�����ʯ��ˮ�У�Ca(OH)2+CO2=CaCO3
+H2O�����ABCD��CaCO3+2HCl=CaCl2+H2O+CO2����������ͨ�����ʯ��ˮ�У�Ca(OH)2+CO2=CaCO3![]() +H2O��
+H2O��
��2��ϡ������лӷ��ԣ�����ƵõĶ�����̼�����лẬ���Ȼ��������ˮ���������ñ��͵�̼��������Һ�ɳ�ȥ�Ȼ������壬Ũ���������ˮ�ԣ�������ˮ��������������и�����ø�װ�þ�������ʱ��������Ҫ�����̳�����ˮ����������һ�������c��d��a��b��
��3��̼�����ϡ���ᷴӦʱ�����ɵ��������һ������ḽ����ʯ��ʯ�ı��棬��ֹ��Ӧ�Ľ�һ�����У����������ϡ�������ϡ���ᡣ������ܣ����ɵ�����Ƹ�����ʯ��ʯ�ı��棬��ֹ��Ӧ�Ľ�һ�����С�
��4���������ܶ�С���ռ������ɲ��������ſ����������Ե�����Ӧ���Ƕ̵ĵ����ܽ����ĵ����ܳ������ ��
��

 ��ʱѵ���������������ϵ�д�
��ʱѵ���������������ϵ�д� �ƸԾ���Ȥζ����ϵ�д�
�ƸԾ���Ȥζ����ϵ�д� ����С����ҵ��ϵ�д�
����С����ҵ��ϵ�д�����Ŀ����� A ��B ��������ѡһ����������������𣬰� A �Ʒ֡�
��һ�������£�Mg �� MgH2���ת������ʵ�������Ĵ�����ͷš��乤��ԭ����ͼ��
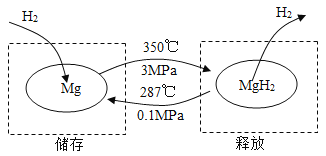
A | B |
��1����������ʱ������Ӧ�Ļ�ѧ����ʽΪ_______�� ��2���������������ݣ��������洢Ч��Ϊ 10%������ 1 kg ������������Ҫ Mg ������Ϊ_______ kg�� | ��1�������ͷ�ʱ������Ӧ�Ļ�ѧ����ʽ Ϊ_______�� ��2�������ϣ�52 kg MgH2������ͷ�����������Ϊ_______ kg�� |