��Ŀ����
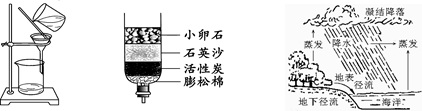
��4�֣�ˮ������֮Դ������Ϊ�˽������������ˮ�ѵ����⣬��2010��ĩ���������ӹ�ˮһ�ڹ��̿�����Ա��ʦ��ᡣ
��1����ˮ�к����������ʣ����������������������˺�����ȷ������������о����̶���ߵķ�����___________��
��2��Ӳˮ����������������ܶ��鷳�������г���______________________������Ӳˮ����ˮ����������еķ���������ˮ��Ӳ�ȣ�
��3������ϴ�ྫϴ�����ۣ�����ϴ�ྫ����___________���ܣ�
��4����84����Һ�������ڶ�ˮԴ������ɱ�������Ʊ�����Ҫ��ӦΪ��
2NaOH + Cl2 =" NaClO" +NaCl+H2O��֤����84����Һ�����Ƿ���NaCl�ķ����ǣ�ȡ������84����Һ������������______________________��Һ�������ƣ����۲��Ƿ����������ϡ����İ�ɫ������
��1����ˮ�к����������ʣ����������������������˺�����ȷ������������о����̶���ߵķ�����___________��
��2��Ӳˮ����������������ܶ��鷳�������г���______________________������Ӳˮ����ˮ����������еķ���������ˮ��Ӳ�ȣ�
��3������ϴ�ྫϴ�����ۣ�����ϴ�ྫ����___________���ܣ�
��4����84����Һ�������ڶ�ˮԴ������ɱ�������Ʊ�����Ҫ��ӦΪ��
2NaOH + Cl2 =" NaClO" +NaCl+H2O��֤����84����Һ�����Ƿ���NaCl�ķ����ǣ�ȡ������84����Һ������������______________________��Һ�������ƣ����۲��Ƿ����������ϡ����İ�ɫ������
��1������ ��2������ˮ ��3���黯 ��4����������Һ��ϡ���ᣨ�������ữ����������Һ����������Һ��
����1������ķ������Եõ���ˮ�����÷����Ǿ����̶���ߵģ��ʱ����Ϊ������
��2��Ӳˮ�к��еĸ�þ���ӽ϶࣬��ˮ�к����ٵĻ��Dz�����þ���ӣ�������ˮ�м������ˮ��������������ĭ���ʿ����÷���ˮ������Ӳˮ����ˮ���ʱ����Ϊ������ˮ
��3��ϴ�ྫ֮������ȥ������������ϴ�ྫ�����黯���ܣ��ʱ����Ϊ���黯
��4�����������ӵķ�������������������Һ����Ϊ�����ܹ����ɳ�����Ϊ���ų�̼������ӵĸ��ţ�ͨ������ϡ�����ữ��Һ���ʱ����Ϊ����������Һ��ϡ����
��2��Ӳˮ�к��еĸ�þ���ӽ϶࣬��ˮ�к����ٵĻ��Dz�����þ���ӣ�������ˮ�м������ˮ��������������ĭ���ʿ����÷���ˮ������Ӳˮ����ˮ���ʱ����Ϊ������ˮ
��3��ϴ�ྫ֮������ȥ������������ϴ�ྫ�����黯���ܣ��ʱ����Ϊ���黯
��4�����������ӵķ�������������������Һ����Ϊ�����ܹ����ɳ�����Ϊ���ų�̼������ӵĸ��ţ�ͨ������ϡ�����ữ��Һ���ʱ����Ϊ����������Һ��ϡ����

��ϰ��ϵ�д�
�����Ŀ