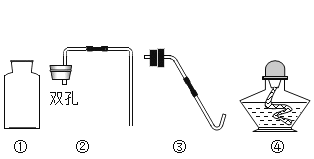
��Ŀ����
����Ŀ����ͭ����ZnΪ��Ҫ����Ԫ�ص�ͭ�Ͻ𡣻�ͭ����Լ��Zn 7%��ZnO 31%��Cu 50%��CuO 5%������Ϊ���ʡ�������ͭ���ɵõ�����п������Ҫ�������£����ʲ�����ˮ��Ҳ�����뷴Ӧ����
��֪��ZnO+H2SO4=ZnSO4+H2O CuO+H2SO4=CuSO4+H2O
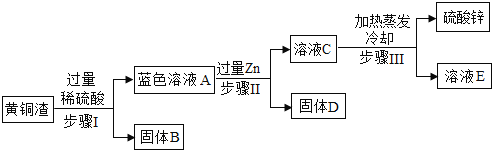
��1��������в�������Ļ�ѧ����ʽ______________________________��
��2����ҺA�еĽ�����������__________________���ѧ�����
��3������������о������IJ�������Ϊ____________________��
��4����ҺA________����������������С������������������ҺC��������
���𰸡� Zn��H2SO4��ZnSO4��H2�� Zn2+��Cu2+ ���� С��
����������1������п������ͭ�ֱ���ϡ���ᷴӦ��������п������ͭ��ˮ��û�������������п��ϡ���ᷴӦ��������п�����������Zn+H2SO4=ZnSO4+H2������2��п��ϡ���ᷴӦ��������п������������п�����ᷴӦ��������п��ˮ������ͭ�����ᷴӦ��������ͭ��ˮ�����еĽ�����������п���Ӻ�ͭ���ӣ����Zn2+��Cu2+����3����ͼ��֪����������˳�ȥͭ�����ʣ�����������dz�ȥп���û�����ͭ�����˺����������о��õ����������ֲ�����������4��A��C�Ĺ����з����ķ�Ӧ�ǣ�Zn+CuSO4=Cu+ZnSO4��
160 161
�ɴ˿ɼ���Һ��������������ҺAС����ҺC��������

 ����������ϵ�д�
����������ϵ�д�