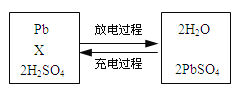
��Ŀ����
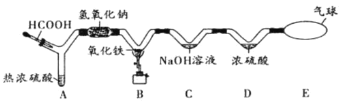
����Ŀ����1��2001���й���Э�Բ���װ������ڻ���״���������Ժ��֣����������ڱ���Ⱦ������������֯�Ѱѱ���Ϊǿ���°����ʡ�����һ��û����ɫ����������ζ��Һ�壬�ܶȱ�ˮС��������ˮ�����ķе���80.1�棬�۵���5.5�档��һ�������£����ֱ��ܸ��������塢Ũ���ᡢŨ��������ʷ�����ѧ��Ӧ���������ڿ�����ȼ�����ɶ�����̼��ˮ����ش��������⣺
��������������____________________________________________��
���Ļ�ѧ������____________________________________________��
���ڿ�����ȼ�յ���������ʽ��_________________________________________��
��2��������Ϣ��д��ѧ����ʽ��ϰ��
�������ǵĻ����������N2H4����ȼ�ϣ���������������N2O4������������ȼ��β���ɵ�����ˮ������ɡ���д����Ӧ�ķ��ű���ʽ_____________________________________��
����ʳ��ԭ�����ʱ�����������Ƶõľ��ᣬ������Ϊ�ƾ���C2H5OH����ø��������������е��������ã������˴��ᣨCH3COOH����ˮ����д���÷�Ӧ����������ʽΪ_________��
���𰸡�û����ɫ����������ζ��Һ�壬�ܶȱ�ˮС��������ˮ�����ķе���80.1�棬�۵���5.5�档 �ܸ��������塢Ũ���ᡢŨ��������ʷ�����ѧ��Ӧ�����п�ȼ�� ��+����![]() ������̼+ˮ ����+����������
������̼+ˮ ����+����������![]() ����+ˮ �Ҵ�+����
����+ˮ �Ҵ�+����![]() ����+ˮ
����+ˮ
��������
�������������У�û����ɫ����������ζ��Һ�壬�ܶȱ�ˮС��������ˮ�����ķе���80.1�棬�۵���5.5�档��
���Ļ�ѧ�����У��ܸ��������塢Ũ���ᡢŨ��������ʷ�����ѧ��Ӧ�����п�ȼ�ԣ�
���ڿ�����ȼ�ղ���������̼��ˮ����������ʽ����+����![]() ������̼+ˮ��
������̼+ˮ��
��2���������ǵĻ����������������������ȼ�շ�Ӧ������ˮ������Ӧ�ķ��ű���ʽ������+����������![]() ����+ˮ��
����+ˮ��
����ʳ��ԭ�����ʱ�����������Ƶõľ��ᣬ�ƾ���ø��������������е��������ã������˴����ˮ����������ʽΪ���Ҵ�+����![]() ����+ˮ��
����+ˮ��

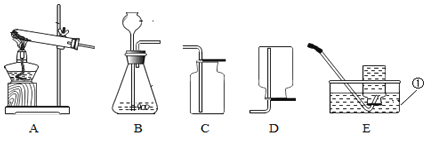
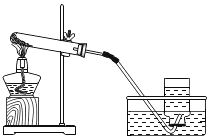
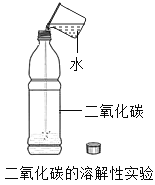
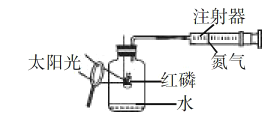
����Ŀ��ijУ��ѧ��ȤС���ͬѧ���������ͼ��ʾ��װ�ý��п�������������ʵ���̽��:
ʵ��ǰע��������20 mL����,100 mL����ƿ�м���10 mLˮ��ʵ�����ݼ�¼����:
ʱ��/min | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
ע��������/mL | 20.0 | 34.0 | 40.0 | 30.0 | 25.6 | 18.0 | 12.5 | 8.6 | 4.8 | 2.0 | 2.0 |
��1����������ʵ����̺������ۺϷ���,����ƿ�ڿ����������������____________mL;��ʼ��������ԭ����_______________��
��2����ʽ����������������������______________��
��3�����������ƫС,ԭ�������________________��
����Ŀ�����ࡢ��ȡ���������ѧϰ��ѧ���õķ���.
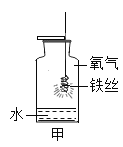
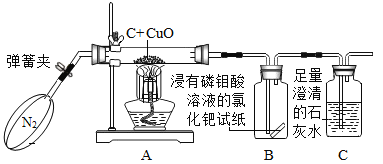
��1�����л�ѧ������ʵ�飬Ϊ�˱����о������ǿ��ѻ�ѧʵ�鰴��̽��Ŀ�ķ�Ϊ̽�����ʵ����ʡ�̽�����ʵ��Ʊ���̽����Ӧ�����ȡ��ݴ�Ӧ������ʵ���е�D��______��Ϊһ�࣬������_________��
|
|
|
|
A | B | C | D |
��2����֪Cl2+H2O=HCl+HClO��Ϊ��̽��������HClO��Ư���ԣ�ijͬѧ��������µ�ʵ�顣
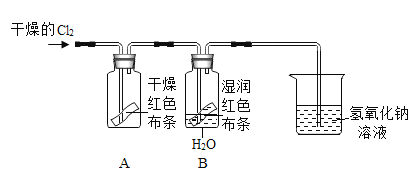
��ͨ��Cl2�Ӽ���ƿA�и���ĺ�ɫ��������ɫ������ɵó��Ľ�����________���Ӽ���ƿB��ʪ��ĺ�ɫ������ɫ������ɵó��Ľ�����__________��
��Ϊ��֤����HClOʹʪ��ĺ�ɫ������ɫ������Ϊ��Ӧ���ӵ�ʵ����________��