��Ŀ����
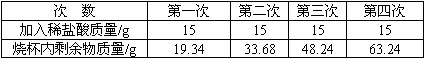
Ϊ�˲ⶨij��ʯ��ʯ��̼��Ƶ�����������ȡ5 gʯ��ʯ��Ʒ�����ձ��У���60 gϡ����ֳ��Ĵμ����ձ��У���ַ�Ӧ���ʵ���������±�(��Ʒ�е����ʲ������ᷴӦҲ������ˮ)
��������������йؼ��㣺
(1)�ļ��η�Ӧ��������ʣ��___________________________________________��
(2)ʯ��ʯ��Ʒ��̼��Ƶ�����������
(3)����ʵ����������ձ��ڵ������м���ʯ��ʯ�����ٲ������ݣ����ˡ�����Һ�����ʵ�����������(�����ȷ��0.1%)
(1)������(1��)
(2)�⣺��5 gʯ��ʯ��CaCO3������Ϊx��
CaCO3��2HCl===CaCl2��H2O��CO2��(1��)
100 44
x 1.76 g(1��)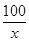 ��
��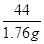 ��x��4 g(1��)
��x��4 g(1��)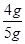 ��100%��80%(1��)
��100%��80%(1��)
(3)��60 g������ȫ��Ӧ��CaCO3����Ϊy�����ɵ�CaCl2����Ϊz��������CO2����Ϊm��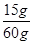 ��
��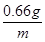 ��m��2.64 g(��0.66��4��2.64 g)(1��)
��m��2.64 g(��0.66��4��2.64 g)(1��)
(��������һ�βμӷ�Ӧ��CaCO3������1.5 g��1.5 g��4��6 g��ͨ����һ�η�Ӧ�����������������������Ϊ7.3%Ҳ��1��)
CaCO3��2HCl===CaCl2��H2O��CO2��
100 111 44
y z 2.64 g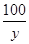 ��
��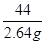 ��y��6 g(1��)
��y��6 g(1��)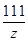 ��
��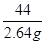 ��z��6.66 g(1��)
��z��6.66 g(1��)
��Һ������6 g��60 g��2.64 g��63.36 g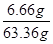 ��100%��10.5%(1��)
��100%��10.5%(1��)
��(��)
(���������������÷�)
����

| �Ρ��� | ��һ�� | �ڶ��� | ������ | ���Ĵ� |
| ����ϡ��������/g | 15 | 15 | 15 | 15 |
| �ձ���ʣ��������/g | 19.34 | 33.68 | 48.24 | 63.24 |
��1���ļ��η�Ӧ��������ʣ��
��2��ʯ��ʯ��Ʒ��̼��Ƶ�����������
��3������ʵ����������ձ��ڵ������м���ʯ��ʯ�����ٲ������ݣ����ˣ�����Һ�����ʵ������������������ȷ��0.1%��