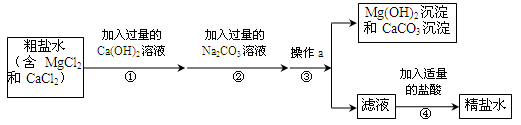
��Ŀ����
������ͼ�ش����⣺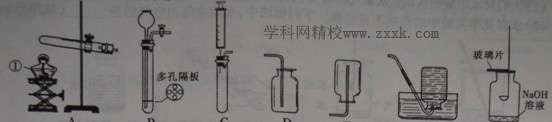
(1)д��ͼ�б���������ƣ��� ��
��2��ʵ�����ø��������ȡ���ռ�����Ӧѡ�õ�װ���� ������ţ���ͬ������Ӧ�Ļ�ѧ����ʽΪ �����װ��������ʱ���������ֽ��յķ����⣬�����Բ��õķ�����
(3)ʵ���ҿ��������ƹ�����Ũ��ˮ�ڳ����»����ȡ����������Ϊ�˵õ�ƽ�ȵİ�������Ӧѡ��ķ���װ���� ��
(4)��Gװ��������������ȼ��ʵ�飬ȼ�ս�����ȡ��ȼ�ճף������ò���Ƭ��סƿ�ڲ�����������ƿ��ת�����ֲ���Ƭ�� ����ס������������(���ͼ)����ԭ���� ��
(5)Ϊ�������ʵ�飬��lOmL��Ͳ��ȡ6mL����������Һ������ʱ��Ͳ�ڵ�Һ�尼Һ����ʹ�Ӧ����ͼ�� ��(�a"��b��)�̶��߱���ˮƽ��
��1���ƾ��� (1��)��
��2��AF����AD�� (1��)��2KMnO4 K2MnO4+ MnO2+O2����2�֣�������ë����ס�Թܣ����þƾ������Թܵȣ� ��1�֣�
K2MnO4+ MnO2+O2����2�֣�������ë����ס�Թܣ����þƾ������Թܵȣ� ��1�֣�
��3��C ��1�֣�
��4��SO2��NaOH��Һ��Ӧ������ƿ����ѹ��С��������ѹ������Ƭ����ס����2�֣�
��5��a ��1�֣�
����

ʵ��������һƿ��ɫҺ�壬ijͬѧ˵��������Һ��Ʒ�еμ�ʯ����Һ���ж�������ԡ��û������ڿ�ѧ̽���ģ� ��
| A��������� | B���������� | C�����ʵ�� | D����˼���� |
ij��ѧС����̽����Ļ�ѧ����ʱ��������ͼ��ʾ������ʵ�飮
��1��д��A�Թ��з�����Ӧ�Ļ�ѧ����ʽ�� ����
��2��ʵ�������ͬѧ�ǽ�A��B���Թ��еķ�Һ����ͬһ�ྻ���ձ����ձ��г�����ɫ������С��ͬѧ���ձ��е����ʽ����˹��ˣ�����Һ�гɷ������²��룺
��������롿�ף���Һ�е�������Na2SO4 CuSO4
�ң���Һ�е�������Na2SO4 H2SO4
������Һ�е��������� 4��
������Һ�е�������Na2SO4
���������ۡ�ͬѧ�Ƕ����ϵIJ���������ɣ���Ϊ������ ������������������
�����ʵ�顿ͬѧ��ͨ�����µ�ʵ��Բ��������֤���������ʵ�鱨�森
| ʵ������ | Ԥ������ | Ԥ�ƽ��� |
| ��1��ȡ������Һ���Թ��У��۲� | ��Һ����ɫ | ������ �������� |
| ��2�������������Թ��м����� �� | ��������ɫ������ | ������ ������ |

��12�֣�С���ڻ�ѧʵ�鼼�ܿ����У���ɡ���ϡ�����������������Һ��̼������Һ������̼������Һ��pH��ʵ�顣
��ʵ��һ��С��ȡ����Һ�ס��ҷֱ�������Թܢٺ͢ڣ�������֧�Թ��е���ϡ���ᣬ�۲�ʵ��������������֪����Ϊ̼������Һ��
��1��ʵ���У�С��Ӧ�۲쵽�Թܢڵ�������__________________________________��
���Թ��з�Ӧ�Ļ�ѧ����ʽΪ_________________________________��
��2����ʦָ�����������⣬����ʹ�������Լ������������ƺ�̼������Һ������ѡ��һ�ֲ���������Լ�����������ʵ���е�Ԥ�������Լ���ѧ����ʽ�����±���
| ѡ����Լ� | Ԥ������ | ��Ӧ�Ļ�ѧ����ʽ |
| ____________ | һ֧�Թ����������� ��һ֧�Թ�________________ | _________________________ |
| ʵ������ | ���� |
| ��Һմ����ֽ��ʱ����ֽ�����ر�ɫ | �����ɫ���Աȣ�Na2CO3��ҺpH="3" |
��_______________________________________________________________��
��4��ָ��С���¼�ġ�ʵ�������в���ѧ�ĵط���
��_______________________________________________��
(9��) һ��Ȥζ��ѧ��У�����ʦ��ͬѧ��չʾ��һƿ��ǩ�������ɫ��Һ����ͼ��ʾ��Ҫ��ͬѧ�ǽ���̽����ȷ����ƿ��Һ������ʲô��Һ��
��������롿 ����ʦ��ʾ����ƿ��ɫ��Һֻ��������������Һ�е�һ�֣�������þ��Һ ����������Һ ��������Һ���������Һ
���������ϡ�
�ٳ����£�������ʵ��ܽ�����£�
| ���� | MgSO4 | Na2SO4 | (NH4)2SO4 | H2SO4 |
| �ܽ�� | 35��1g | 19��5g | 75��4g | ��ˮ����Ȼ��� |
��ʵ��̽������1��С��������ʦ����ʾ���������� ��ԭ���� ��
��2��Ϊȷ���������ֲ����Ƿ���ȷ��С��ͬѧ��������̽����
| ʵ����� | ʵ������ | ʵ����� |
| ��ȡ����Һ�������Թ��У������еμӼ��� ��Һ | ��Һ���а�ɫ�������� | ����ٳ��� |
| ���ò�����պȡ����ԭ��Һ����pH��ֽ�ϣ���������ɫ������ | ��ҺpHС��7 | ����۳��� |
��3���������ʵ�鷽����ȷ�ϸ���Һ���������Һ�����ʵ�鱨�棺
| ʵ����� | ʵ������ | ʵ����� |
| ȡ����Һ�������Թ��У� | | ����ܳ������÷�Ӧ�Ļ�ѧ����ʽΪ |