��Ŀ����
Ϊ�˲ⶨijţ����Ʒ�е����ʵĺ������ֲ��á��Ƕ�����ֽ����еĵ����ʣ���ԭ���ǰѵ������еĵ�Ԫ����ȫת���ɰ�������ѧʽ��NH3��������ϡ�������հ�������Ӧ�Ļ�ѧ����ʽΪ��2NH3+H2SO4�T��NH4��2SO4����ȡ��ţ����Ʒ30mL�á��Ƕ�����ֽ����еĵ����ʣ������İ�����9.5g������������Ϊ4.9%��ϡ����ǡ����ȫ���գ����㲢�ش��������⣺��1�����������������Ƕ��ٿˣ�����������ȷ��0.01g����ͬ��
��2��30mLţ���к���Ԫ�ص������Ƕ��ٿˣ�
������д����ѧ����ʽ���������ݼ����������������Ȼ����������غ㶨�ɿ�֪�������е�Ԫ�ص��������ڵ������е�Ԫ�ص�������
����⣺���������������9.5��4.9%=0.4655�ˣ��谱����������X��
2NH3+H2SO4�T��NH4��2SO4
34 98
X 0.4655
�T
��X=0.16
�ఱ���е�Ԫ�ص���������0.16��
��100%�T0.13g��
����������غ㶨�ɿ�֪���������е�Ԫ�ص��������ڰ����еĵ�Ԫ�ص�������
��30mLţ���к���Ԫ�ص�������0.13�ˣ�
�𣺲���������������0.16�ˣ�30mLţ���к���Ԫ�ص�������0.13�ˣ�
2NH3+H2SO4�T��NH4��2SO4
34 98
X 0.4655
| 34 |
| 98 |
| X |
| 0.4655 |
��X=0.16
�ఱ���е�Ԫ�ص���������0.16��
| 14 |
| 17 |
����������غ㶨�ɿ�֪���������е�Ԫ�ص��������ڰ����еĵ�Ԫ�ص�������
��30mLţ���к���Ԫ�ص�������0.13�ˣ�
�𣺲���������������0.16�ˣ�30mLţ���к���Ԫ�ص�������0.13�ˣ�
���������ջ�ѧ����ʽ�ļ��㷽���淶�ԣ����������غ㶨�ɵ�Ӧ�ã�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
 Ϊ�˲ⶨijţ����Ʒ�е����ʵĺ������ֲ��á��Ƕ�����ֽ����еĵ����ʣ���ԭ���ǰѵ������еĵ�Ԫ����ȫת���ɰ�������ѧʽΪ��NH3��������ϡ�������հ�������Ӧ�Ļ�ѧ����ʽ��
Ϊ�˲ⶨijţ����Ʒ�е����ʵĺ������ֲ��á��Ƕ�����ֽ����еĵ����ʣ���ԭ���ǰѵ������еĵ�Ԫ����ȫת���ɰ�������ѧʽΪ��NH3��������ϡ�������հ�������Ӧ�Ļ�ѧ����ʽ��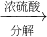 �����
�����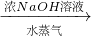 ����Ϊ�˲ⶨijţ����Ʒ�е����ʵĺ�����ȡ30mLţ���øǶ�����뵰���ʣ��ѵ�Ԫ����ȫת���ɰ���NH3������50g4.9%ϡH2SO4��Һ���պ�ʣ�������Ҫ��38.0g4.0%��NaOH��Һ�кͣ���
����Ϊ�˲ⶨijţ����Ʒ�е����ʵĺ�����ȡ30mLţ���øǶ�����뵰���ʣ��ѵ�Ԫ����ȫת���ɰ���NH3������50g4.9%ϡH2SO4��Һ���պ�ʣ�������Ҫ��38.0g4.0%��NaOH��Һ�кͣ���