��Ŀ����
����Ŀ����������װ��ͼ���ش��й�����:
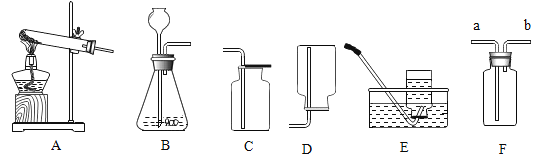
(1)��װAװ��ʱ���ƾ���Ӧ�ڹ̶��Թ�֮____(�ǰ����)���ã�Bװ���г���©����������Һ�����µ�ԭ����_____��
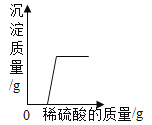
(2)�ӽ��ܻ������濼�ǣ�ʵ������ȡ�ϴ�����O2��Ӧ��ͼ��ѡ��_____(����)������ϣ�������Ӧ�Ļ�ѧ����ʽΪ_____��
(3)ʵ������ȡCO2��Ӧ�Ļ�ѧ����ʽΪ_____��ʵ������Է�Ӧ��IJ���������������̲���:
![]()
����ʱ�����õIJ����������ձ��⣬����_____��(����������)�������������У���_____ʱ��ֹͣ���ȣ����������������ʹ��Һ���ɡ�
(4)����Fװ�ý����ſ������ռ�H2��H2��Fװ�õ�_____(�a����b��)��ͨ�롣
���𰸡�ǰ ��ֹ���ɵĶ�����̼�ӳ���©���ݳ� BE 2H2O2![]() 2H2O+O2�� CaCO3+2HCl=CaCl2+H2O+CO2�� ©���������� �������г��ֽ϶������� b
2H2O+O2�� CaCO3+2HCl=CaCl2+H2O+CO2�� ©���������� �������г��ֽ϶������� b
��������
��1����װAװ��ʱ���ƾ���Ӧ�ڹ̶��Թ�֮ǰ���ã����ݻ����λ��ȷ���Թܸ߶ȣ�Bװ���г���©����������Һ�����µ�ԭ���ǣ���ֹ���ɵ�����ӳ���©���ݳ���
��2�������˫��ˮ�Ͷ��������������Ͳ���Ҫ���ȣ��ȽϽ��ܻ�����������������ˮ������ˮ��Ӧ������ˮ���ռ��������Ƚϴ�������ѡBE�����������ڶ��������������������·�Ӧ��������ʽΪ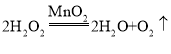 ��
��
��3��ʵ�����ô���ʯ��ϡ������ȡ������̼�ķ���ʽΪCaCO3+2HCl=CaCl2+H2O+CO2������Ӧ��������õIJ����������ձ��⣬����©���Ͳ������������������У����������г��ֽ϶�������ʱ��ֹͣ���ȣ�
��4������Fװ�ý����ſ������ռ�H2����Ϊ�������ܶȱȿ���С��H2��Fװ�õĶ̹ܽ��룬�����ӳ����ų������b��