��Ŀ����
����ˮ�೧�����ң�Ϊ�˲ⶨij��ɽʯ��ʯ��̼��Ƶ�����������ȡʯ��ʯ��Ʒ������ϡ�������ձ��з�Ӧ������ʯ��ʯ��Ʒ�����ʲ���ϡ���ᷴӦҲ������ˮ�����й�ʵ���������±���
| | ��Ӧǰ | ��Ӧ�� | |
| ʵ������ | �ձ���ϡ��������� | ʯ��ʯ��Ʒ������ | �ձ������л��������� |
| 160 g | 12 g | 167.6 g | |
��2�����ʯ��ʯ��̼��Ƶ�����������
������1�����������غ㶨�ɣ�������̼������Ϊ��160 g+12 g��167.6 g = 4.4g
��2���⣺���ʯ��ʯ��Ʒ��̼��Ƶ�����Ϊx��
CaCO3+2HCl=CaCl2+H2O+CO2��
100 44
x 4.4 g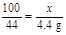

��ʯ��ʯ��̼��Ƶ���������Ϊ��  ��(��)
��(��)
�������ڽ������ʱ�����ȷ������п�������⣬Ȼ����ѧ����֪ʶ�����е�֪ʶ���з������

��ϰ��ϵ�д�
 ��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�
��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�
�����Ŀ
����ˮ�೧�����ң�Ϊ�˲ⶨij��ɽʯ��ʯ��̼��Ƶ�����������ȡʯ��ʯ��Ʒ������ϡ�������ձ��з�Ӧ������ʯ��ʯ��Ʒ�����ʲ���ϡ���ᷴӦҲ������ˮ�����й�ʵ���������±���
��1�����������غ㶨�ɿ�֪����Ӧ�����ɶ�����̼������Ϊ g��
��2�����ʯ��ʯ��̼��Ƶ�����������
| ��Ӧǰ | ��Ӧ�� | ||
| ʵ������ | �ձ���ϡ��������� | ʯ��ʯ��Ʒ������ | �ձ������л��������� |
| 160g | 12g | 167.6g | |
��2�����ʯ��ʯ��̼��Ƶ�����������
����ˮ�೧�����ң�Ϊ�˲ⶨij��ɽʯ��ʯ��̼��Ƶ�����������ȡʯ��ʯ��Ʒ������ϡ�������ձ��з�Ӧ������ʯ��ʯ��Ʒ�����ʲ���ϡ���ᷴӦҲ������ˮ�����й�ʵ���������±���
��1�����������غ㶨�ɿ�֪����Ӧ�����ɶ�����̼������Ϊ g��
��2�����ʯ��ʯ��̼��Ƶ�����������
| | ��Ӧǰ | ��Ӧ�� | |
| ʵ������ | �ձ���ϡ��������� | ʯ��ʯ��Ʒ������ | �ձ������л��������� |
| 160 g | 12 g | 167.6 g |
��2�����ʯ��ʯ��̼��Ƶ�����������
����ˮ�೧�����ң�Ϊ�˲ⶨij��ɽʯ��ʯ��̼��Ƶ�����������ȡʯ��ʯ��Ʒ������ϡ�������ձ��з�Ӧ������ʯ��ʯ��Ʒ�����ʲ���ϡ���ᷴӦҲ������ˮ�����й�ʵ���������±���
|
|
��Ӧǰ |
��Ӧ�� |
|
|
ʵ������ |
�ձ���ϡ��������� |
ʯ��ʯ��Ʒ������ |
�ձ������л��������� |
|
160 g |
12 g |
167.6 g |
��1�����������غ㶨�ɿ�֪����Ӧ�����ɶ�����̼������Ϊ g��
��2�����ʯ��ʯ��̼��Ƶ�����������