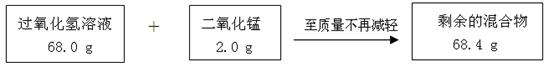
��Ŀ����
��ͼ�Dz�����Һʱ�õ�һƿ������ˮ��ǩ������ݱ�ǩ���������ݼ��㣺

��1����ƿ��Һ�к�ˮ g��
��2������ÿ����60�Σ�20��ԼΪ1mL�������ʸ����˾�����Һ500mL��Լ�� �������ꡣ
(3)ijѧϰС��Ϊ�˼����ǩ˵���Ƿ���ʵ����������ʵ�����ȡ100g����Һ�������м������������������г���������AgNO3 + NaCl="==" AgCl�� +NaNO3�����Ƶó���������Ϊ2.2g��ͨ������˵����ǩ˵���Ƿ���ʵ������ȷ��0.1%��

��1����ƿ��Һ�к�ˮ g��
��2������ÿ����60�Σ�20��ԼΪ1mL�������ʸ����˾�����Һ500mL��Լ�� �������ꡣ
(3)ijѧϰС��Ϊ�˼����ǩ˵���Ƿ���ʵ����������ʵ�����ȡ100g����Һ�������м������������������г���������AgNO3 + NaCl="==" AgCl�� +NaNO3�����Ƶó���������Ϊ2.2g��ͨ������˵����ǩ˵���Ƿ���ʵ������ȷ��0.1%��
��45.5 ��167 ��3��0.9%
��1����������������ʽ�ɼ������Һ����������Һ������ȥNaCl�����������Ǹ�ƿ��Һ�к�ˮ��������
��2��60��20�ɼ������ÿ������Һ�������500ml����ÿ������Һ�����������Ҫ��ʱ�䣮
�Ǹ����Ȼ��ƺ�������������ѧ��Ӧ�ķ���ʽ�����100����Һ�е��Ȼ��Ƶ��������ٽ�һ�������Һ���ֵܳ���������������������ˮ�ı�ǩ�Աȵó�����
�ǽ�:���Ȼ�����Һ�����ʵ�����ΪX
NaCl + AgNO3 ="===" AgCl�� + NaNO3
58.5 143.5
X 2.2g
 X=0.897g
X=0.897g
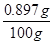 ��100��=0.9��
��100��=0.9��
����������Ϊ0.9����������ʵ��
��2��60��20�ɼ������ÿ������Һ�������500ml����ÿ������Һ�����������Ҫ��ʱ�䣮
�Ǹ����Ȼ��ƺ�������������ѧ��Ӧ�ķ���ʽ�����100����Һ�е��Ȼ��Ƶ��������ٽ�һ�������Һ���ֵܳ���������������������ˮ�ı�ǩ�Աȵó�����
�ǽ�:���Ȼ�����Һ�����ʵ�����ΪX
NaCl + AgNO3 ="===" AgCl�� + NaNO3
58.5 143.5
X 2.2g
 X=0.897g
X=0.897g 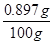 ��100��=0.9��
��100��=0.9������������Ϊ0.9����������ʵ��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ