��Ŀ����
��ֽ���ҹ��Ŵ��Ĵ���֮һ������Ч���ƶ������������ķ�չ��
��1������ũ����Ľո���Ϊ��ֽԭ�ϣ����Լ��ٷ��սոѴ������̳���Ⱦ���ոѵ���Ҫ �ɷ�֮һ����ά��[��ѧʽ ��C6H10O5��n]������������������Ԫ���� �� ��ά������ ��ѡ��л����������������֮һ����
��2����ֽ�����л����������NaOH�ķ�ˮ��Сӱͬѧ����pH��ֽ���Բⶨ��ˮ���ȣ��ⶨ�ľ��巽���� ��
��3��Ϊ�����÷�ˮ�Ի�������Ⱦ�����ϡ���ᡢ����ͭ��̼�������ҩƷ��ѡ��һ��ҩƷ������ȥ��ˮ�е�NaOH������д���ó��ӷ�Ӧ�Ļ�ѧ����ʽ�� ��
��1������ũ����Ľո���Ϊ��ֽԭ�ϣ����Լ��ٷ��սոѴ������̳���Ⱦ���ոѵ���Ҫ �ɷ�֮һ����ά��[��ѧʽ ��C6H10O5��n]������������������Ԫ����
��2����ֽ�����л����������NaOH�ķ�ˮ��Сӱͬѧ����pH��ֽ���Բⶨ��ˮ���ȣ��ⶨ�ľ��巽����
��3��Ϊ�����÷�ˮ�Ի�������Ⱦ�����ϡ���ᡢ����ͭ��̼�������ҩƷ��ѡ��һ��ҩƷ������ȥ��ˮ�е�NaOH������д���ó��ӷ�Ӧ�Ļ�ѧ����ʽ��
���㣺Ԫ�ص�������������,��Һ�����Ȳⶨ,�л��������������,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺��ѧʽ�ļ���,��ѧ����������غ㶨��,���ʵķ���,������������ѧʵ���������
��������1�����ݻ�ѧʽ�и�Ԫ�ص������Ⱥ��л���ĸ�����������
��2��������Һ�ļ��Գ������ָʾ����pH��ֽ��
��3����������к͵�ԭ����������
��2��������Һ�ļ��Գ������ָʾ����pH��ֽ��
��3����������к͵�ԭ����������
����⣺��1������ά��[��C6H10O5��n]�У�̼Ԫ�ص�����Ϊ��12��6��n=72n����Ԫ�ص�����Ϊ��1��10��n=10n����Ԫ�ص�����Ϊ��16��5��n=80n���ɼ�������Ԫ�ص��������������ά����һ�ֺ�̼Ԫ�صĻ���������л��
��2��������Һ�ļ��Գ������ָʾ����PH��ֽ�������÷�̪��Һ���м��飬�����֤����Һ�ʼ��ԣ�
��3��ѡ�����ᣬ�����ˮ�е��������Ʒ����кͷ�Ӧ�����������ƺ�ˮ��
�ʴ�Ϊ����1����Ԫ�أ��л��
��2����pH��ֽ���ڽྻ����İ״�Ƭ�������ϣ��ýྻ����IJ�����պȡ����Һ������pH��ֽ�ϣ�Ѹ�������ɫ�����ж��գ�
��3��2NaOH+H2SO4=Na2SO4+2H2O��
��2��������Һ�ļ��Գ������ָʾ����PH��ֽ�������÷�̪��Һ���м��飬�����֤����Һ�ʼ��ԣ�
��3��ѡ�����ᣬ�����ˮ�е��������Ʒ����кͷ�Ӧ�����������ƺ�ˮ��
�ʴ�Ϊ����1����Ԫ�أ��л��
��2����pH��ֽ���ڽྻ����İ״�Ƭ�������ϣ��ýྻ����IJ�����պȡ����Һ������pH��ֽ�ϣ�Ѹ�������ɫ�����ж��գ�
��3��2NaOH+H2SO4=Na2SO4+2H2O��
������������Ҫ���黯ѧʽ�ĺ��弰Ӧ������к͵�ԭ�����ʵ�������������

��ϰ��ϵ�д�
 ���ٴ�����ɽ����ϵ�д�
���ٴ�����ɽ����ϵ�д�
�����Ŀ
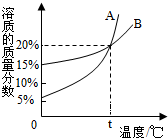 ��ͼ��A��B�������ʵı�����Һ�����ʵ������������¶ȱ仯�����ߣ��ֽ�20gA��40gB�ֱ�����100gˮ�У�����ʹ����ȫ�ܽ⣬Ȼ������ȴ��t�棬�����й���������ȷ���ǣ�������
��ͼ��A��B�������ʵı�����Һ�����ʵ������������¶ȱ仯�����ߣ��ֽ�20gA��40gB�ֱ�����100gˮ�У�����ʹ����ȫ�ܽ⣬Ȼ������ȴ��t�棬�����й���������ȷ���ǣ�������| A��t��ʱ��A��Һ�����ʵ���������С��B |
| B��t��ʱ��A��Һ�����ʵ�������������B |
| C��t��ʱ������Һ�о������������� |
| D��t��ʱ�����ߵ��ܽ����� |
���Ƕ��ѡ���ڳ��⣬����Ⱦơ���������ˬ�ı��֣�����������ʳ������ɹ���������ɣ�Ҳ��������ʳ�������ѪҺ��pHȴԼΪ7.35���������ֺ�ˬ�Խ����������ȾƳ����ͬʱ������������������еģ�������
| A���մ�ˮ | B������ | C����ˮ | D����֭ |
��A��B��C��D���ֽ������ֱ�����Ǽ��뵽ϡ�����У�A��B��D��Ӧ��������ų���C�����������ٰ�A��D����B����Һ�У�D������B��������Aû�У������ֽ����Ļ��˳����Ϊ��������
| A��A��B��D��C |
| B��B��D��A��C |
| C��D��A��B��C |
| D��D��B��A��C |
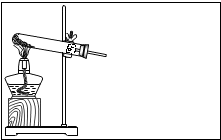 ��ͼ��С��ͬѧ���ڻ��Ƶ�ʵ�����ø��������ȡ����������ˮ���ռ������װ��ͼ����ش��������⣺
��ͼ��С��ͬѧ���ڻ��Ƶ�ʵ�����ø��������ȡ����������ˮ���ռ������װ��ͼ����ش��������⣺