��Ŀ����
����һֱ��̽������İ��ء������Ϻ��� H��He��N��Na��Mg��Si ��Ԫ�أ�δ�����ܻ��Ϊ����ı�����Դ��������ͼ�ش�
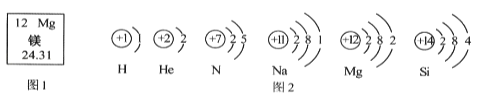
��1��þԭ�ӵ����ԭ��������_____________ ��
��2��He�Ǹ�Ч����ԭ�ϣ���ԭ�ӵĺ����������________________��
��3��H Ԫ�غ�HeԪ�صı���������_____________��ͬ��
��4������ͼ2��Ϣ����ѧ֪ʶ����������˵������ȷ����______________��
A H�Ƿǽ���Ԫ��
B Nԭ�����������Ӳ�
C He�� Mg�Ļ�ѧ��������
��5��SiO2��һ��____________�������ʷ��ࣩ�����������ά������SiԪ�صĻ��ϼ���______��
��6������һ�ֻ��ý�������ˮ��Ӧ�Ļ�ѧ����ʽΪ2Na + 2H2O = 2NaOH + H2��,�÷�Ӧ�Ļ�����Ӧ������_______________��NaOH��Һ�������ӵķ�����___________��
��ϰ��ϵ�д�
�����Ŀ
��������ʵ������д��Ӧ�����ԭ����
ʵ�� |
�õ�ñ����ƾ��� |
�ø�ѹˮǹ��ˮ��� |
��ֹ�����ӣ�����������ľ |
ԭ�� | ____ | ____ | ____ |