��Ŀ����
����ij�������ŷŵķ�ˮ�к���HCl������ԱС��Ա����ŷŵķ�ˮ��HCl�������������вⶨ����20mL��ˮ���ٶ����ܶ�Ϊ1g/cm3����Ʒ������ƿ�У�����������Ϊ1%������������Һ�����кͣ�����ȥ����������Һ8g�������ˮ�в���������������������Һ��Ӧ�����ʣ���
��1������÷�ˮ�к�HCl������������
��2���ó�ÿ�������ˮ100�֣���ֱ���ŷŽ��Ի��������Ⱦ���������������������ܽ����ŷţ��������������Ʒ�ĩ����������Ϊ10%������������Һ�����кͣ��й��������±���ʾ��
| ���� | ���ʣ������ʣ�����Է������� | �г��ο��ۣ�Ԫ/�֣� |
| �������Ʒ�ĩ | 74 | 200 |
| 10%������������Һ | 40 | 100 |
�⣺��1����ó��ŷŵķ�ˮ�к�HCl����������Ϊx
NaOH+HCl�TNaCl+H20
40 36.5
8g��1% 20mL��1g/cm3��x
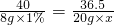
x=0.365%
�𣺸ó��ŷŵķ�ˮ�к�HCl����������Ϊ0.365%��
��2�������������Ƹ�����ķ�Ӧ��ѧ����ʽ����ϣ�1���Ľ�������ɸ���Ľ��
��20ml��ˮ���ĵ��������Ƶ�����Ϊy��
Ca��OH��2+2HCl=CaCl2+2H2O
74 73
y 20g��0.365%
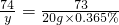
y=0.074g
��100t��ˮ���ĵ��������Ƶ�����Ϊ0.37t����Ҫ�ʽ�74Ԫ�����ݣ�1���Ľ��20ml��ˮ����1%������������Һ0.08g����ô����10%������������ҺΪ0.8g����������֪��100t��ˮ����10%���������Ƶ�����Ϊ��4t����Ҫ�ʽ�400Ԫ������Ӧ��ѡ������������������ˮ��
�ʴ�Ϊ��
��1��0.365%��
��2��Ca��OH��2���������Ƶ���Է�������Ϊ74�����ݼ���ֱ��к�100�ַ�ˮ�������������Ʒ�ĩO��37����Ҫ�ʽ�74Ԫ��������������Ϊ10%����������Һ4����Ҫ�ʽ�400Ԫ���������ɱ�����ѡ���������Ʒ�ĩ��
��������1�����ݡ��������Ƶ�����=������������������������뷴Ӧ���������Ƶ�������Ȼ������������������ᷴӦ�Ļ�ѧ����ʽ���������Ƶ����������ɼ�������뷴Ӧ���������������������Ȼ��������������
��2�����Ը����������ơ��������Ʒֱ������ᷴӦ�Ļ�ѧ����ʽ������������ƺ��������Ƶ�������Ȼ����бȽϣ�
������������Ҫ����ѧ�����û�ѧ����ʽ�����ʵ�����������ʽ���м�����������ѶȽϴ�
NaOH+HCl�TNaCl+H20
40 36.5
8g��1% 20mL��1g/cm3��x
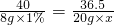
x=0.365%
�𣺸ó��ŷŵķ�ˮ�к�HCl����������Ϊ0.365%��
��2�������������Ƹ�����ķ�Ӧ��ѧ����ʽ����ϣ�1���Ľ�������ɸ���Ľ��
��20ml��ˮ���ĵ��������Ƶ�����Ϊy��
Ca��OH��2+2HCl=CaCl2+2H2O
74 73
y 20g��0.365%
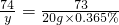
y=0.074g
��100t��ˮ���ĵ��������Ƶ�����Ϊ0.37t����Ҫ�ʽ�74Ԫ�����ݣ�1���Ľ��20ml��ˮ����1%������������Һ0.08g����ô����10%������������ҺΪ0.8g����������֪��100t��ˮ����10%���������Ƶ�����Ϊ��4t����Ҫ�ʽ�400Ԫ������Ӧ��ѡ������������������ˮ��
�ʴ�Ϊ��
��1��0.365%��
��2��Ca��OH��2���������Ƶ���Է�������Ϊ74�����ݼ���ֱ��к�100�ַ�ˮ�������������Ʒ�ĩO��37����Ҫ�ʽ�74Ԫ��������������Ϊ10%����������Һ4����Ҫ�ʽ�400Ԫ���������ɱ�����ѡ���������Ʒ�ĩ��
��������1�����ݡ��������Ƶ�����=������������������������뷴Ӧ���������Ƶ�������Ȼ������������������ᷴӦ�Ļ�ѧ����ʽ���������Ƶ����������ɼ�������뷴Ӧ���������������������Ȼ��������������
��2�����Ը����������ơ��������Ʒֱ������ᷴӦ�Ļ�ѧ����ʽ������������ƺ��������Ƶ�������Ȼ����бȽϣ�
������������Ҫ����ѧ�����û�ѧ����ʽ�����ʵ�����������ʽ���м�����������ѶȽϴ�

��ϰ��ϵ�д�
�����Ŀ
�����н�����ĺ����ߡ������غ�̲Ϳ�����ĺ�����������غ��ˣ����γ��ˡ���ijУѧ���Դ�Ϊ����չ������о���
��1���γ��ڹŴ���ʢ�����Ρ���������ͨ����ɹ��ˮ�����ξ�ˮ�ȣ����Եõ����Σ�
�ٺ�ˮ��ɹ�Ρ��õ��Ĵ����к�����������ɳ�Ȳ��������ʣ���ͨ�����в�����ȡ���Σ��ܽ�����ˡ� ����д�������裩��������ι��˺���Һ�Ի��ǣ�ԭ������� ��д��һ�ּ��ɣ���
�ڴ����ᴿʵ���У��ȳ�ȡ5.0g���Σ���ȡ10mLˮֻ�ܽ�4.0g���Σ����յõ�3.6g���Σ����εIJ����� ��
��2���������̲����ḻ��ʯ����Ȼ�����зḻ���غ�������Դ����糡�Ľ���Ͷ���������ڼ���CO2���ŷţ�
����Ȼ���Dz���������Դ������Ҫ�ɷ�Ϊ �����ѧʽ��
��CO2���ŷ�����ɡ�����ЧӦ����ԭ��֮һ������˵����ȷ���� ����дѡ����ţ���
A��ֲ�����֣��Ͻ��ҿ��ķ���ľ
B����Լ��Դ������ú��ʯ�͵Ȼ�ʯȼ�ϵ�ȼ��
C���Ͻ��ֲġ����ġ����Ͻ��������ȸ��ܺIJ�ҵ������
D�������¼���������̫���ܡ����ܡ����ܡ������ܵ�����Դ
��3�����غ����ô��Ľ����У�Ҫ��ֿ����뻷�����أ�
��ij�������мס����������䣬�ų��ķ�ˮ���������ֱ������ֲ�ͬ�����ӣ�����K+��Ba2+��Cu2+��OH-��NO3-��CO32-�������ӣ��������ʵ��ܽ��Լ��±�������֪�׳����ˮ���Գʼ��ԣ����ҳ����ˮ�к��е��������� �����ס���������ķ�ˮ���ʵ�������ϣ����Ա��Ϊ��������ʹ��ˮ�е�Ba2+��Cu2+��OH-������ת��Ϊ������ȥ���ֿ������ϲ���ҹ����ũ���ҹ�е�������Ҫ�� ����д���ʻ�ѧʽ����
�ڸ����ϱ��жϣ�Cu��NO3��2��Ba��OH��2��Һ�ܲ��ܷ������ֽⷴӦ���ܷ�Ӧд����ѧ����ʽ�����ܷ�Ӧд�����ɣ� ��
��1���γ��ڹŴ���ʢ�����Ρ���������ͨ����ɹ��ˮ�����ξ�ˮ�ȣ����Եõ����Σ�
�ٺ�ˮ��ɹ�Ρ��õ��Ĵ����к�����������ɳ�Ȳ��������ʣ���ͨ�����в�����ȡ���Σ��ܽ�����ˡ�
�ڴ����ᴿʵ���У��ȳ�ȡ5.0g���Σ���ȡ10mLˮֻ�ܽ�4.0g���Σ����յõ�3.6g���Σ����εIJ�����
��2���������̲����ḻ��ʯ����Ȼ�����зḻ���غ�������Դ����糡�Ľ���Ͷ���������ڼ���CO2���ŷţ�
����Ȼ���Dz���������Դ������Ҫ�ɷ�Ϊ
��CO2���ŷ�����ɡ�����ЧӦ����ԭ��֮һ������˵����ȷ����
A��ֲ�����֣��Ͻ��ҿ��ķ���ľ
B����Լ��Դ������ú��ʯ�͵Ȼ�ʯȼ�ϵ�ȼ��
C���Ͻ��ֲġ����ġ����Ͻ��������ȸ��ܺIJ�ҵ������
D�������¼���������̫���ܡ����ܡ����ܡ������ܵ�����Դ
��3�����غ����ô��Ľ����У�Ҫ��ֿ����뻷�����أ�
��ij�������мס����������䣬�ų��ķ�ˮ���������ֱ������ֲ�ͬ�����ӣ�����K+��Ba2+��Cu2+��OH-��NO3-��CO32-�������ӣ��������ʵ��ܽ��Լ��±�������֪�׳����ˮ���Գʼ��ԣ����ҳ����ˮ�к��е���������
| ������������ | OH- | NO3- | CO32- |
| K+ | �� | �� | �� |
| Ba2+ | �� | �� | �� |
| Cu2+ | �� | �� | �� |
�����н�����ĺ����ߡ������غ�̲Ϳ�����ĺ�����������غ��ˣ����γ��ˡ���ijУѧ���Դ�Ϊ����չ������о���
��1���γ��ڹŴ���ʢ�����Ρ���������ͨ����ɹ��ˮ�����ξ�ˮ�ȣ����Եõ����Σ�
�ٺ�ˮ��ɹ�Ρ��õ��Ĵ����к�����������ɳ�Ȳ��������ʣ���ͨ�����в�����ȡ���Σ��ܽ�����ˡ�______����д�������裩��������ι��˺���Һ�Ի��ǣ�ԭ�������______��д��һ�ּ��ɣ���
�ڴ����ᴿʵ���У��ȳ�ȡ5.0g���Σ���ȡ10mLˮֻ�ܽ�4.0g���Σ����յõ�3.6g���Σ����εIJ�����______��
��2���������̲����ḻ��ʯ����Ȼ�����зḻ���غ�������Դ����糡�Ľ���Ͷ���������ڼ���CO2���ŷţ�
����Ȼ���Dz���������Դ������Ҫ�ɷ�Ϊ______�����ѧʽ��
��CO2���ŷ�����ɡ�����ЧӦ����ԭ��֮һ������˵����ȷ����______����дѡ����ţ���
A��ֲ�����֣��Ͻ��ҿ��ķ���ľ
B����Լ��Դ������ú��ʯ�͵Ȼ�ʯȼ�ϵ�ȼ��
C���Ͻ��ֲġ����ġ����Ͻ��������ȸ��ܺIJ�ҵ������
D�������¼���������̫���ܡ����ܡ����ܡ������ܵ�����Դ
��3�����غ����ô��Ľ����У�Ҫ��ֿ����뻷�����أ�
��ij�������мס����������䣬�ų��ķ�ˮ���������ֱ������ֲ�ͬ�����ӣ�����K+��Ba2+��Cu2+��OH-��NO3-��CO32-�������ӣ��������ʵ��ܽ��Լ��±�������֪�׳����ˮ���Գʼ��ԣ����ҳ����ˮ�к��е���������______�����ס���������ķ�ˮ���ʵ�������ϣ����Ա��Ϊ��������ʹ��ˮ�е�Ba2+��Cu2+��OH-������ת��Ϊ������ȥ���ֿ������ϲ���ҹ����ũ���ҹ�е�������Ҫ��______����д���ʻ�ѧʽ����
�ڸ����ϱ��жϣ�Cu��NO3��2��Ba��OH��2��Һ�ܲ��ܷ������ֽⷴӦ���ܷ�Ӧд����ѧ����ʽ�����ܷ�Ӧд�����ɣ�______��
��1���γ��ڹŴ���ʢ�����Ρ���������ͨ����ɹ��ˮ�����ξ�ˮ�ȣ����Եõ����Σ�
�ٺ�ˮ��ɹ�Ρ��õ��Ĵ����к�����������ɳ�Ȳ��������ʣ���ͨ�����в�����ȡ���Σ��ܽ�����ˡ�______����д�������裩��������ι��˺���Һ�Ի��ǣ�ԭ�������______��д��һ�ּ��ɣ���
�ڴ����ᴿʵ���У��ȳ�ȡ5.0g���Σ���ȡ10mLˮֻ�ܽ�4.0g���Σ����յõ�3.6g���Σ����εIJ�����______��
��2���������̲����ḻ��ʯ����Ȼ�����зḻ���غ�������Դ����糡�Ľ���Ͷ���������ڼ���CO2���ŷţ�
����Ȼ���Dz���������Դ������Ҫ�ɷ�Ϊ______�����ѧʽ��
��CO2���ŷ�����ɡ�����ЧӦ����ԭ��֮һ������˵����ȷ����______����дѡ����ţ���
A��ֲ�����֣��Ͻ��ҿ��ķ���ľ
B����Լ��Դ������ú��ʯ�͵Ȼ�ʯȼ�ϵ�ȼ��
C���Ͻ��ֲġ����ġ����Ͻ��������ȸ��ܺIJ�ҵ������
D�������¼���������̫���ܡ����ܡ����ܡ������ܵ�����Դ
��3�����غ����ô��Ľ����У�Ҫ��ֿ����뻷�����أ�
��ij�������мס����������䣬�ų��ķ�ˮ���������ֱ������ֲ�ͬ�����ӣ�����K+��Ba2+��Cu2+��OH-��NO3-��CO32-�������ӣ��������ʵ��ܽ��Լ��±�������֪�׳����ˮ���Գʼ��ԣ����ҳ����ˮ�к��е���������______�����ס���������ķ�ˮ���ʵ�������ϣ����Ա��Ϊ��������ʹ��ˮ�е�Ba2+��Cu2+��OH-������ת��Ϊ������ȥ���ֿ������ϲ���ҹ����ũ���ҹ�е�������Ҫ��______����д���ʻ�ѧʽ����
| ������������ | OH- | NO3- | CO32- |
| K+ | �� | �� | �� |
| Ba2+ | �� | �� | �� |
| Cu2+ | �� | �� | �� |