��Ŀ����
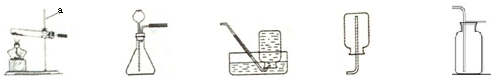
��ͼ��ʾ��ʵ��������ȡ����ʱ����װ�á���ش��������⣺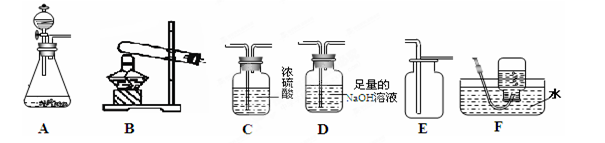
��1����ʵ��������ȡ����ʱ������ҩƷ֮ǰһ��Ҫ�ȼ��� ��
��2����ʯ��ʯ��ϡ����Ϊԭ����ȡ���ռ������CO2���塣
�ٸ�װ�õ���ȷ����˳���ǣ���װ����ţ� ��
�ڲ���CO2����Ļ�ѧ����ʽΪ ��
��Cװ�õ������� ��
�ܼ��鼯��ƿ���Dz���CO2����ķ��� ��
��3��ͬѧ��Ϊ��֤��ȡCO2��ʣ����Һ���Ƿ������ᣨ���ʲ�����ˮ�Ҳ��μӷ�Ӧ����CaCl2��Һ�����ԣ������ʵ�鷽�����£�ȡ�����Թ��У��μ�AgNO3��Һ��������ɫ�������ټ���ϡ���ᣬ�������ܽ⣬�������ᡣ���Ƿ�ͬ��÷������������ɡ� ��
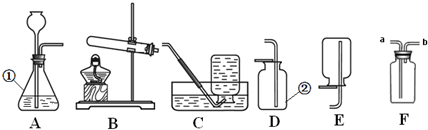
��4��������CO2����ͨ��ʢ������Ca(OH)2��Һ�Ĺ��ƿ�У�ƿ����Һ��������֮ǰ��Ȼ� ����д���и�����ţ���
| A������ | B����С | C������ | D�����ж� |
��1��װ�õ�������
��2����ACE����CaCO3+2HCl=CaCl2+2H2O+CO2�����۸�����������������е�ˮ�֣��ܼ��������ij���ʯ��ˮ���������������ʯ��ˮ����ǣ�����CO2��
��3����ͬ�⣬��Ϊʣ����Һ��һ������CaCl2�������Ƿ���ϡ���ᣬ�μ�AgNO3�������������ϡ����İ�ɫ������
��4��A
���������������1����ʵ������ȡ����ʱ������ҩƷ֮ǰһ��Ҫ�ȼ���װ�õ������ԣ��Է����ɵ������ݳ�����2����ʵ�����ô���ʯ��ϡ���ᷴӦ��ȡ������̼����Ӧ���״�ǹ����Һ�壬��Ӧ�����dz��£�Ӧѡ�÷���װ��A�����ڶ�����̼���ܶȴ��ڿ������ܶȣ���ѡ���ռ�װ��B�����ռ�ǰ��Ũ������и�����Կɲ��õ�װ�������ACE����ʵ������ȡCO2�ô���ʯ��ϡ���ᣬ��ѧ����ʽΪ��CaCO3 +2HCl�TCaCl2+H2O+CO2������Ũ���������ˮ�ԣ����Կ�����������������������е�ˮ�֣��ܼ���CO2����ķ������������ij���ʯ��ˮ���������������ʯ��ˮ����ǣ�����CO2��
��3����Һ��һ������CaCl2�������Ƿ���ϡ���ᣬ���ܺ�AgNO3��Һ��Ӧ���ɲ�����ϡ����İ�ɫ��������˸÷��������ƣ�
��4��������̼�ܺ��������Ʒ�Ӧ����̼���ƺ�ˮ����˽�����CO2����ͨ��ʢ������NaOH��Һ�Ĺ��ƿ�У�ƿ����Һ��������֮ǰ��Ȼ����ӣ���ѡA
���㣺��������ķ���װ�ú��ռ�װ����ѡȡ������֤������Ϳ�����������

��ͼ���ռ�����R���岢��ˮ�����������մ�������ȷװ�á���ͼ��ʵ��װ���Ʋ��������й����ʣ���ȷ��һ����
| | A | B | C | D |
| �ܶȣ�������Ƚϣ� | ���� | ���� | С�� | С�� |
| ��ˮ�е��ܽ��� | ���� | ������ | ������ | ���� |
��5�֣�����ѧϰ�˳��������ʵ�����Ʒ���֪������ȡ�����һ��˼·�ͷ�����
��1��ʵ������ȡ����ʱ��ȷ������װ����Ҫ���ǵ������� ������ĸ��ţ���
| A����Ӧ���״̬ | B��������ܶ� |
| C����Ӧ������ | D��������ܽ��� |

����a�������� ��
���������ȡ����ѡ�õķ���װ���� ������ĸ��ţ�����װ��C�ռ�������̼��ԭ���� ��
�۳�����̼���ƹ�����ˮ��Ӧ�����Ƶ���Ȳ���壬��֪��Ȳ���ܶ�������ӽ���������ˮ��ʵ��������ȡ��Ȳ��ѡ���װ���� ������ĸ��ţ���