��Ŀ����
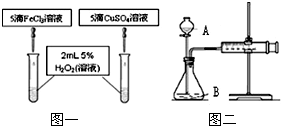
����MnO2��CuSO4��Һ������H2O2��Һ�ֽ�Ĵ�����ijУ��ѧ��ȤС����̽������һЩ���������������Һ�Ƿ�Ҳ������H2O2��Һ�ֽ�Ĵ���������������ǵ�̽�����̣���1��[С���IJ���]��Al2O3������������ֽ�Ĵ�����
[С���IJ���]��FeCl3��Һ������������ֽ�Ĵ�����
��2��[ʵ����֤]��
| ʵ�鲽�� | ʵ������ | ʵ����� | |
| С����ʵ�� | �������ǵ�ľ������װ�й���������Һ���Թ��� | ľ������ȼ | |
| | ľ����ȼ | Al2O3�ܼӿ�H2O2��Һ�ķֽ����� | |
| С����ʵ�� | ��һ֧�Թ��м���H2O2��Һ��Ȼ�����������FeCl3��Һ���Ѵ����ǵ�ľ�������Թܣ� | | FeCl3��Һ�ӿ�H2O2��Һ�ķֽ����� |
��4��[��˼1]���е�ͬѧ��ΪС����ʵ�飬������ȫ֤��Al2O3��H2O2��Һ�ֽ������˴����ã���Ӧ������̽��Al2O3 ��
[��˼2]����֪FeCl3��ˮ�пɽ����Fe3+��Cl-��ͬѧ��������²��룺
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�H2O��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Fe3+��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Cl-��
����Ϊ����ܵ��� ͬѧ�IJ��룬������
��5��ͬѧ�Ƕ����µ��������룬��ʵ�������̽����������ϸ�����������
| ʵ����� | ʵ������ | ���� |
| ��ʢ��5ml5%��H2O2��Һ���Թ��м������� ��HCl�����Ѵ����ǵ�ľ�������Թܣ� | ���������� | |
| ��ʢ��5ml5%��H2O2��Һ���Թ��м������� �� �����Ѵ����ǵ�ľ�������Թܣ� |
��4�����ݴ����Ķ�������ɽ�𣻸��ݹ���������Һ�к���ˮ����������ܵIJ��룻
��5������ʵ�����1���Ը���ʵ�������������ۣ�
ʵ�����2Ϊ����֤Fe3+�Ե�H2O2�Ĵ��ֽ⣬����H2O2��Һ�м���������Fe��NO3��3��Һ��Ȼ�����ʵ�������������ۼ��ɣ�
����⣺��2��[ʵ����֤]С����ʵ����̽��Al2O3�Թ�������ֽ����ʵ�Ӱ�죮ʵ��1�IJ����ǣ��������ǵ�ľ������װ�й���������Һ���Թ��У������ǵ�ľ������ȼ���ɼ������¹�������ֽ����ʺ�����Ȼ����װ�й���������Һ���Թ��м�������Al2O3�������ǵ�ľ����ȼ���ɼ���Al2O3�ܼӿ��������ķֽ����ʣ�
С����ʵ������֤FeCl3��Һ�Թ�������ֽ����ʵ�Ӱ�죬ͨ��ʵ����ۿ���֪�����Ȼ�����Һ�ӿ��˹�������ķֽ����ʣ�
��4�����ݴ����Ķ������֪������֤��һ������Ϊ������������֤�ܷ�ı��������ʵķ�Ӧ�ٶ��⣬������֤�������ͻ�ѧ����Ҳû�з����ı䣻
[��˼2]���ɹ���������Һ�к���ˮ��֪������ܵ��Ǽ�ͬѧ�IJ��룬�����ǹ���������Һ����ˮ��
��5������H2O2��Һ���Թ��м���������HCl��Һ������ʹ�����ǵ�ľ����ȼ��֪��FeCl3��Һ�е�Cl-���ܴ��ֽ�H2O2��
Ϊ����֤Fe3+��H2O2�Ĵ��ֽ⣬����ʢ��5mL��15%��H2O2��Һ���Թ��м���������Fe��NO3��3��Һ���Թ����д������ݲ����������ǵ�ľ����ȼ����Ӧ���ۣ�������õ���Fe3+��
�ʴ�Ϊ����2��[ʵ����֤]��
| ʵ�鲽�� | ʵ������ | ʵ����� | |
| С����ʵ�� | �������ǵ�ľ������װ�й���������Һ���Թ��� | ľ������ȼ | �����¹�������ֽ����ʺ��� |
| װ�й���������Һ���Թ��м�������Al2O3 Ȼ�����ǵ�ľ�������Թ��� | ľ����ȼ | Al2O3�ܼӿ�H2O2��Һ�ķֽ����� | |
| С����ʵ�� | ��һ֧�Թ��м���H2O2��Һ��Ȼ�����������FeCl3��Һ���Ѵ����ǵ�ľ�������Թܣ� | Ѹ�ٲ����������� ľ����ȼ | FeCl3��Һ�ӿ�H2O2��Һ�ķֽ����� |
[��˼2]���ף�����������Һ����ˮ��
��5��
| ʵ����� | ʵ������ | ���� |
| ��ʢ��5ml5%��H2O2��Һ���Թ��м������� ��HCl�����Ѵ����ǵ�ľ�������Թܣ� | ���������� | Cl-���ܴ��ֽ�H2O2 |
| ��ʢ��5ml5%��H2O2��Һ���Թ��м������� �� Fe��NO3��3��Һ�����Ѵ����ǵ�ľ�������Թܣ� | �Թ����д������ݲ��������ǵ�ľ����ȼ | ������õ���Fe3+ |
�������������ص���Ը���Ϊ��һ������䡱��һ�����ܹ��ı仯ѧ��Ӧ���ʣ���������ָ�����ͻ�ѧ�����ڻ�ѧ��Ӧǰ�ֲ��䣮Ҫ̽��һ�������ܷ���Ϊ��ѧ��Ӧ�Ĵ���������ͬʱ�߱������ص㣮

 ������ȫ�̼����ĩ���100��ϵ�д�
������ȫ�̼����ĩ���100��ϵ�д�����MnO2��CuSO4��Һ������H2O2��Һ�ֽ�Ĵ�����ijУ��ѧ��ȤС����̽������һЩ���������������Һ�Ƿ�Ҳ������H2O2��Һ�ֽ�Ĵ���������������ǵ�̽�����̣�
��1��[С���IJ���]��Al2O3������������ֽ�Ĵ�����
[С���IJ���]��FeCl3��Һ������������ֽ�Ĵ�����
��2��[ʵ����֤]��
| ʵ�鲽�� | ʵ������ | ʵ����� | |
| С����ʵ�� | �������ǵ�ľ������װ�й���������Һ���Թ��� | ľ������ȼ | ______ |
| ______ ______ | ľ����ȼ | Al2O3�ܼӿ�H2O2��Һ�ķֽ����� | |
| С����ʵ�� | ��һ֧�Թ��м���H2O2��Һ��Ȼ�����������FeCl3��Һ���Ѵ����ǵ�ľ�������Թܣ� | ______ ______ | FeCl3��Һ�ӿ�H2O2��Һ�ķֽ����� |
��4��[��˼1]���е�ͬѧ��ΪС����ʵ�飬������ȫ֤��Al2O3��H2O2��Һ�ֽ������˴����ã���Ӧ������̽��Al2O3______��
[��˼2]����֪FeCl3��ˮ�пɽ����Fe3+��Cl-��ͬѧ��������²��룺
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�H2O��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Fe3+��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Cl-��
����Ϊ����ܵ���______ ͬѧ�IJ��룬������______
��5��ͬѧ�Ƕ����µ��������룬��ʵ�������̽����������ϸ�����������
| ������ ʵ����� | ʵ������ | ���� |
| ��ʢ��5ml5%��H2O2��Һ���Թ��м������� ��HCl�����Ѵ����ǵ�ľ�������Թܣ� | ���������� | ______ |
| ��ʢ��5ml5%��H2O2��Һ���Թ��м������� ��______�����Ѵ����ǵ�ľ�������Թܣ� | ______ | ______ |
����MnO2��CuSO4��Һ������H2O2��Һ�ֽ�Ĵ�����ijУ��ѧ��ȤС����̽������һЩ���������������Һ�Ƿ�Ҳ������H2O2��Һ�ֽ�Ĵ���������������ǵ�̽�����̡�
��1��[С���IJ���]��Al2O3������������ֽ�Ĵ�����
[С���IJ���]��FeCl3��Һ������������ֽ�Ĵ�����
��2��[ʵ����֤]��
| ʵ�鲽�� | ʵ������ | ʵ����� | |
| С����ʵ�� | �������ǵ�ľ������װ�й���������Һ���Թ��� | ľ������ȼ |
|
|
| ľ����ȼ | Al2O3�ܼӿ�H2O2��Һ�ķֽ����� | |
| С����ʵ�� | ��һ֧�Թ��м���H2O2��Һ��Ȼ�����������FeCl3��Һ���Ѵ����ǵ�ľ�������Թܡ� |
| FeCl3��Һ�ӿ�H2O2��Һ�ķֽ����� |
��3��[����]����Al2O3��FeCl3��Һ����H2O2��Һ�ֽ�Ĵ�����
��4��[��˼1]���е�ͬѧ��ΪС����ʵ�飬������ȫ֤��Al2O3��H2O2��Һ�ֽ������˴����á���Ӧ������̽��Al2O3 ��
[��˼2]����֪FeCl3��ˮ�пɽ����Fe3+ ��Cl-��ͬѧ��������²��룺
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�H2O��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Fe3+��
��ͬѧ�IJ����ǣ��������ֽ�H2O2����FeCl3��Һ�е�Cl-��
����Ϊ����ܵ��� ͬѧ�IJ��룬 ������
��5��ͬѧ�Ƕ����µ��������룬��ʵ�������̽����������ϸ�����������
| ʵ����� | ʵ������ | ���� |
| ��ʢ��5ml5%��H2O2��Һ���Թ��м������� ��HCl�����Ѵ����ǵ�ľ�������Թܡ� | ���������� |
|
| ��ʢ��5ml5%��H2O2��Һ���Թ��м������� �� �����Ѵ����ǵ�ľ�������Թܡ� |
|
|
 ����Уʵ����ʵʩ��������2011���п���ѧʵ���������ո������Ļ��ʵ�鿼����H2O2�ֽ�������Ϊ�β�����MnO2��������ͬѧ��Ϊ֮չ����̽����
����Уʵ����ʵʩ��������2011���п���ѧʵ���������ո������Ļ��ʵ�鿼����H2O2�ֽ�������Ϊ�β�����MnO2��������ͬѧ��Ϊ֮չ����̽����