��Ŀ����
����Ŀ����������������������������ء�
��1����������ͭ������㷺ʹ�õ����ֽ���,������ģ�������������ֽ������Ⱥ�����Ϊ����Ԫ�ط��ű�ʾ��______________��
��2����ͼ��Ԫ�����ڱ���һ��,����˵����ȷ������______��
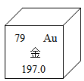
A��ԭ�ӵĺ��������Ϊ118 B������ԭ��������197.0
C��Ԫ�����ڽ���Ԫ�� D��ԭ�ӵ�������Ϊ 97
��3�������������������ڹ���һЩ�����ߺ�

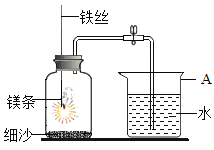
�ٱ������������Ĵ�ʩ֮һ�Ƿ�ֹ����ʴ���������������õķ��ⷽ����______________�������ԭ����________________��
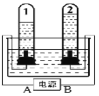
�ڹ���������Щ�������ɸ����Ƴɵġ�ͼ2�ǹ�ҵ�����ķ�Ӧԭ��ģ��ʵ�飬�������е�������________________��������Ӧ�Ļ�ѧ����ʽ��____________ ���ӻ����Ƕȿ�����װ�õIJ���֮����_________________��
��4��ȡͭп�Ͻ��ĩ20g�����ձ��У�����������ϡ���ᣬ��ȫ��Ӧ����������0.2g�������úϽ���ͭ����������______________��
���𰸡�Cu��Fe��Al BC Ϳ�� ����������ˮ ����ɫ�����ɺ�ɫ 3CO+Fe2O3![]() 2Fe+3CO2 û��β������װ�� 67.5%
2Fe+3CO2 û��β������װ�� 67.5%
��������
��1�����������ã�����ͭ���ã����������ģ�������������ֽ������Ⱥ�����˳����Cu��Fe��Al��
��2��A������Ԫ�����ڱ��е�һ���л�ȡ����Ϣ����֪��Ԫ�ص�ԭ������Ϊ79����ԭ���У�ԭ������=������=��������������Խ�ԭ�Ӻ�����79�����ӣ��ʴ���
B������Ԫ�����ڱ��е�һ���л�ȡ����Ϣ����ԭ�ӵ����ԭ������Ϊ197.0������ȷ��
C������Ԫ�����ڱ��е�һ���л�ȡ����Ϣ�����������ڽ���Ԫ�أ�����ȷ��
D������Ԫ�����ڱ��е�һ���л�ȡ����Ϣ����֪��Ԫ�ص�ԭ������Ϊ79�����������������ʴ���
��3�������г�������������Ϳ�͵ķ������⣻�����ԭ���Ǹ���������ˮ��
��һ����̼���л�ԭ�ԣ���������һ����̼������������Ӧ�������Ͷ�����̼���������е������Ǻ���ɫ�����ɺ�ɫ��������Ӧ�Ļ�ѧ����ʽ��3CO+Fe2O3![]() 2Fe+3CO2�����ӻ����Ƕȿ�����װ�õIJ���֮���ǣ�û��β������װ�ã�
2Fe+3CO2�����ӻ����Ƕȿ�����װ�õIJ���֮���ǣ�û��β������װ�ã�
��4����п������Ϊx��
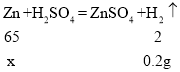
![]() ��
��
x=6.5g��
�Ͻ���ͭ����������=![]() ��
��

����Ŀ����һ�ܱ������У���X��O2��CO2��H2O�������ʣ���һ�������·���ij�ַ�Ӧ����Ӧһ��ʱ���÷�Ӧǰ������ʵ��������±�.����˵����ȷ����
���� | X | O2 | CO2 | H20 |
��Ӧǰ������/g | 19 | 34 | 6 | 2 |
��Ӧ�������/g | 11 | ���� | 28 | 20 |
A. ���еĴ���ֵΪ32 B. X��һ������C��H��OԪ��
C. �÷�Ӧ���û���Ӧ D. ��Ӧ��CO2��H2O��������Ϊ11:9