��Ŀ����
��֪Na2CO3��ˮ��Һ�ʼ��ԣ���һ�ձ���ʢ��20.4g Na2CO3��NaCl��ɵĹ�������������μ������ʷ���Ϊ10%��ϡ���ᣮ�ų��������������������ϡ�����������ϵ������ͼ��ʾ�����������ش����⣺��1�����μ�ϡ������ͼ��B��ʱ���ձ�����Һ��pH______7�����=��������
��2�����μ�ϡ������ͼ��A��ʱ���ձ���Ϊ��������Һ�����£���ͨ����������������ʵ�����������������������һλС����
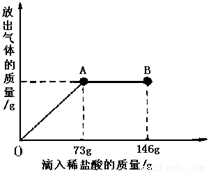
���𰸡�����������ͼʾ���з��������μ�ϡ���ᵽA��ʱ��̼������ȫ��Ӧ����Ӧ���������Ȼ��ƣ�ԭ���Ĺ���������Ҳ���Ȼ��ƣ����������Ȼ����������������������е�̼���Ƶ��������Ӷ�����������������Ȼ��Ƶ��������ټ��Ϸ�Ӧ�����ɵ��Ȼ��Ƶ��������Ƿ�Ӧ�����ʵ�����������Ӧ����Һ������Ϊ�����������ʵ�������ȥ���ɵ������������
����⣺��1����ͼ��֪�����μ�ϡ������A��ʱ��������ȫ��Ӧ�����Ե��μӵ�B��ʱ������������ձ�����Һ��PH��7����2����μ�ϡ���ᵽAʱ���ų����������ΪX���μӷ�Ӧ��̼���Ƶ�����ΪY�����ɵ��Ȼ��Ƶ�����ΪZ
�������֪�����μӵ�Aʱ�������Ȼ����������73g×10%�T7.3g
Na2CO3+2HCl�T2NaCl+H2O+CO2��
106 73 117 44
Y 7.3g Z X
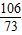 =
= Y�T10.6g
Y�T10.6g
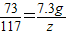 Z�T11.7g
Z�T11.7g
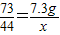 X�T4.4g
X�T4.4g
����Ϊ�Ȼ��ƣ��Ȼ��Ƶ�����Ϊ��11.7g+20.4g-10.6g�T21.5g
��Һ������Ϊ��20.4g+73g-4.4g�T89g
�������ʵ���������Ϊ�� ×100%�T24.2%
×100%�T24.2%
�ʴ�Ϊ����1��������2�����μ�ϡ������ͼ��A��ʱ�����ʵ���������Ϊ24.2%��
�����������ڼ���ʱ����Һ�����ļ����ǹؼ������׳�����Ӧ���������غ㣬��Ӧ����Һ������=����������+����ϡ��������-�ų�����������
����⣺��1����ͼ��֪�����μ�ϡ������A��ʱ��������ȫ��Ӧ�����Ե��μӵ�B��ʱ������������ձ�����Һ��PH��7����2����μ�ϡ���ᵽAʱ���ų����������ΪX���μӷ�Ӧ��̼���Ƶ�����ΪY�����ɵ��Ȼ��Ƶ�����ΪZ
�������֪�����μӵ�Aʱ�������Ȼ����������73g×10%�T7.3g
Na2CO3+2HCl�T2NaCl+H2O+CO2��
106 73 117 44
Y 7.3g Z X
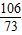 =
= Y�T10.6g
Y�T10.6g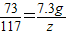 Z�T11.7g
Z�T11.7g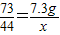 X�T4.4g
X�T4.4g����Ϊ�Ȼ��ƣ��Ȼ��Ƶ�����Ϊ��11.7g+20.4g-10.6g�T21.5g
��Һ������Ϊ��20.4g+73g-4.4g�T89g
�������ʵ���������Ϊ��
 ×100%�T24.2%
×100%�T24.2%�ʴ�Ϊ����1��������2�����μ�ϡ������ͼ��A��ʱ�����ʵ���������Ϊ24.2%��
�����������ڼ���ʱ����Һ�����ļ����ǹؼ������׳�����Ӧ���������غ㣬��Ӧ����Һ������=����������+����ϡ��������-�ų�����������

��ϰ��ϵ�д�
 ��1����Ԫ�¿�������ĩϵ�д�
��1����Ԫ�¿�������ĩϵ�д�
�����Ŀ
ij��ȤС��ͬѧΪ֤��NaOH��Һ��ϡ���ᷢ�����кͷ�Ӧ���Ӳ�ͬ�Ƕ����������ʵ�鷽����������ʵ�飮
����һ������pH��ֽ�ⶨNaOH��Һ��pH���ٵμ����ᣬ����������Һ��ͬʱ�ⶨ�����Һ��pH�������õ�pH��С��С��7����֤��NaOH��Һ��ϡ���ᷢ���˻�ѧ��Ӧ��
��1����pH��ֽ�ⶨ��Һ��pHʱ����ȷ�IJ����ǣ� ��
��2������ǿ������õ�pHС��7�������ɣ� ��
������������NaOH��Һ�еμӼ��η�̪��Һ����Һ�Ժ�ɫ��Ȼ���ٵμ����ᣬ�ɹ۲쵽��ɫ����ʧ����֤��NaOH��Һ��ϡ���ᷢ���˻�ѧ��Ӧ��
����ͬѧ����NaOH��Һ�еμӷ�̪��Һʱ��������һ��������������������Һ�е����̪��Һ����Һ����˺�ɫ������һ�����ɫ����ʧ�ˣ���С����������������ԭ���������²��룺�ٿ����Ƿ�̪��Һ������е�������Ӧ��ʹ��ɫ��ʧ���ڿ���������������Һ������еĶ�����̼��Ӧ��ʹ��ɫ��ʧ��
��1��Ϊ��֤����٣�����ͬѧ��������ʵ�飺�����Ƶ�����������Һ���ȣ�����Һ���Ϸ���һЩֲ���ͣ�Ȼ������ȴ�����Һ�е����̪��Һ��ʵ���С����ȡ��͡�����ֲ���͡�Ŀ���� ��ʵ����������̪��Һ��ɫ��ʧ������е������أ�
��2��Ϊ��֤����ڣ�����ͬѧ��������ʵ�飺ȡ��һ������Na2CO3��Һ�������е����̪��Һ��������ҺҲ���ֺ�ɫ���ɴ˿ɵó�����������ۣ�����1��˵��Na2CO3��Һ�� �ԣ�����2��˵����̪��Һ��ɫ��ʧ������еĶ�����̼�أ�
��3����С��ͬѧͨ���������ϵ�֪��������������ҺŨ�Ⱥܴ�ʱ���ͻ���������������������ʵ��֤���÷�����ȡ�õ�NaOH��ҺŨ�ȹ���ʵ�鷽�� ���ڹ۲쵽������ ��
����������ѧ��Ӧ��ͨ�������������ı仯���ɽ�����Ӧǰ����¶ȱ仯���жϷ�Ӧ�ķ��������NaOH��Һ��ϡ������ǰ���¶��б仯����֤�������˻�ѧ��Ӧ��
����ͬѧ����ͬŨ�ȵ������NaOH��Һ��10mL��ϣ�����
�ȼƲⶨ�����»��ǰ���¶ȵı仯������¼��ÿ�λ��ǰ���¶ȵ�����ֵ��t���������
��1������x= ��
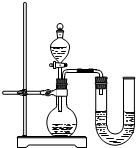
��2��ijͬѧ��ûʹ���¶ȼƵ�����£�ͨ����ͼ��ʾװ�������ʵ�飮���ͬѧ���� �ж�NaOH��Һ��ϡ���ᷢ�����кͷ�Ӧ��
��3������ʵ���е�ϸ�ں������������ʵ���У�ϡ��������ý�ͷ�ι���εμӣ���������Ŀ���� ����ʵ������У�Ҫ�ò��������Ͻ��裬��������Ŀ���� ������ʵ����������ⷢ�������ݳ��֣�����Ϊԭ���� ���ܷ���ʢ��NaOH��Һ���Լ�ƿƿ�ں���Ƥ���ϳ����˰�ɫ��ĩ������������ѧ�Ļ�ѧ֪ʶ�������ְ�ɫ��ĩ�ijɷ��������²��룺�ٿ�����NaOH������Ϊ���ڿ����� �� �ۿ����� ��
��4��Ϊ�˽�һ���о�ʵ���г��ֵ����⣬ȡ��13.3g�������ƹ�����Ʒ��������ˮ�����Һ�������м���200g10%��ϡ���ᣬʹ���ַ�Ӧ�����ɶ�����̼2.2g����
��1����Ʒ���������Ƶ�������
��2�����������Ʒ�Ӧ�������������
��3����ͼ�л������������ʾ������̼�������������ʾ����������Ĺ�ϵͼ��
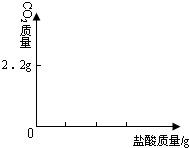
����֪Na2CO3+2HCl�T2NaCl+H2O+CO2����
����һ������pH��ֽ�ⶨNaOH��Һ��pH���ٵμ����ᣬ����������Һ��ͬʱ�ⶨ�����Һ��pH�������õ�pH��С��С��7����֤��NaOH��Һ��ϡ���ᷢ���˻�ѧ��Ӧ��
��1����pH��ֽ�ⶨ��Һ��pHʱ����ȷ�IJ����ǣ�
��2������ǿ������õ�pHС��7�������ɣ�
������������NaOH��Һ�еμӼ��η�̪��Һ����Һ�Ժ�ɫ��Ȼ���ٵμ����ᣬ�ɹ۲쵽��ɫ����ʧ����֤��NaOH��Һ��ϡ���ᷢ���˻�ѧ��Ӧ��
����ͬѧ����NaOH��Һ�еμӷ�̪��Һʱ��������һ��������������������Һ�е����̪��Һ����Һ����˺�ɫ������һ�����ɫ����ʧ�ˣ���С����������������ԭ���������²��룺�ٿ����Ƿ�̪��Һ������е�������Ӧ��ʹ��ɫ��ʧ���ڿ���������������Һ������еĶ�����̼��Ӧ��ʹ��ɫ��ʧ��
��1��Ϊ��֤����٣�����ͬѧ��������ʵ�飺�����Ƶ�����������Һ���ȣ�����Һ���Ϸ���һЩֲ���ͣ�Ȼ������ȴ�����Һ�е����̪��Һ��ʵ���С����ȡ��͡�����ֲ���͡�Ŀ����
��2��Ϊ��֤����ڣ�����ͬѧ��������ʵ�飺ȡ��һ������Na2CO3��Һ�������е����̪��Һ��������ҺҲ���ֺ�ɫ���ɴ˿ɵó�����������ۣ�����1��˵��Na2CO3��Һ��
��3����С��ͬѧͨ���������ϵ�֪��������������ҺŨ�Ⱥܴ�ʱ���ͻ���������������������ʵ��֤���÷�����ȡ�õ�NaOH��ҺŨ�ȹ���ʵ�鷽��
����������ѧ��Ӧ��ͨ�������������ı仯���ɽ�����Ӧǰ����¶ȱ仯���жϷ�Ӧ�ķ��������NaOH��Һ��ϡ������ǰ���¶��б仯����֤�������˻�ѧ��Ӧ��
����ͬѧ����ͬŨ�ȵ������NaOH��Һ��10mL��ϣ�����
�ȼƲⶨ�����»��ǰ���¶ȵı仯������¼��ÿ�λ��ǰ���¶ȵ�����ֵ��t���������
| ��� | ���� | NaOH��Һ | ��t/�� |
| 1 | 3.65% | 2.00% | 3.5 |
| 2 | 3.65% | 4.00% | x |
| 3 | 7.30% | 8.00% | 14 |

��1������x=
��2��ijͬѧ��ûʹ���¶ȼƵ�����£�ͨ����ͼ��ʾװ�������ʵ�飮���ͬѧ����
��3������ʵ���е�ϸ�ں������������ʵ���У�ϡ��������ý�ͷ�ι���εμӣ���������Ŀ����
��4��Ϊ�˽�һ���о�ʵ���г��ֵ����⣬ȡ��13.3g�������ƹ�����Ʒ��������ˮ�����Һ�������м���200g10%��ϡ���ᣬʹ���ַ�Ӧ�����ɶ�����̼2.2g����
��1����Ʒ���������Ƶ�������
��2�����������Ʒ�Ӧ�������������
��3����ͼ�л������������ʾ������̼�������������ʾ����������Ĺ�ϵͼ��

����֪Na2CO3+2HCl�T2NaCl+H2O+CO2����
 �ᡢ������й㷺��;����Ҫ�����ij��ѧ�С���ͬѧΧ���⼸����������һϵ�е�̽�����
�ᡢ������й㷺��;����Ҫ�����ij��ѧ�С���ͬѧΧ���⼸����������һϵ�е�̽�����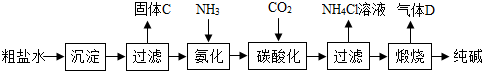

 ��2011?�����ʼ죩���������ƹ����п��ܻ���̼���ƣ��ס��ҡ�����λͬѧ�ֱ�ȡ������ˮ���м��飮
��2011?�����ʼ죩���������ƹ����п��ܻ���̼���ƣ��ס��ҡ�����λͬѧ�ֱ�ȡ������ˮ���м��飮