��Ŀ����
��7�֣�ͭ�������������õĽ���֮һ�� ұ��ͭ����ͭ��ʯ������ȸʯ������Ҫ�ɷ�Ϊ��ʽ̼��ͭ[Cu2(OH)2CO3]����ʽ̼��ͭ��һ����ɫ�IJ�����ˮ�Ĺ��壬�����ֽ⣬������Ϸ��������汸���̼���εĻ�ѧ���ʣ����������ᷢ����Ӧ��
���ͬѧ����ʵ�������Լ�ʽ̼��ͭ�����ۡ�ϡ����Ϊԭ������ȡͭ�����ʵ���������£�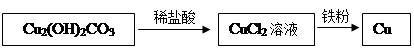
��ش��������⣺
��1����ʽ̼��ͭ�к��� ��Ԫ�ء�
��2����ʽ̼��ͭ���ȷֽ��������ֳ����������������һ��ͨ���Ǻ�ɫ���壬��д���ú�ɫ������ľ̿������Ӧ�Ļ�ѧ����ʽ ����ʽ̼��ͭ��ϡ���ᷴӦ�Ļ�ѧ����ʽΪCu2(OH)2CO3+4HCl====2CuCl2+CO2��+3H2O��Ԥ��ʵ������ ��
��3������ȸʯ��Ʒ�к���44.4gCu2(OH)2CO3����ַ�Ӧ���������ܵõ�ͭ������Ϊ g��
��(1) 4��1�֣�����2��2CuO + C ����2Cu + CO2��(2��)����ɫ��������ʧ���д������ݲ�������Һ����ɫ����ɫ(2��)����������㼴�����֣���3��25.6��2�֣�.
����

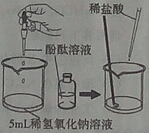
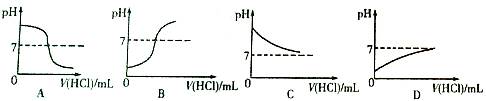
�����ܽ���ѧϰ��ѧ����Ҫ������С��ͬѧ��ͼ1�ܽ���NaOH��������ѧ���ʣ���NaOH�����������ܹ�������ѧ��Ӧ����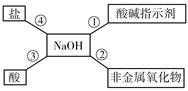
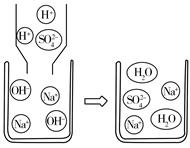
ͼ1 ͼ2
��1����֤��Ӧ�٣�С������ɫ��̪��Һ����NaOH��Һ�У���Һ��� ��
��2��С������ʾ��ͼ2��˵����Ӧ�۵ķ������û�ѧ��Ӧ����ʽ��ʾΪ ���Ӹ�ͼ�п��Կ�������Ӧ�����ҺpH 7�����������������=������
��3�������Ӧ���ܹ���������ѡ��������� ��
| A��Na2CO3 | B��HCl | C��CuSO4 | D��NaCl |