��Ŀ����
 ��6.5��п��������ϡ���ᷴӦ��
��6.5��п��������ϡ���ᷴӦ����1������H2������Ϊ���٣�
��2������H2�ڱ�״��������ı���ʽΪ
��3����������п��245��ϡ���ᷴӦ�����������������Ƕ��٣����������ʱ������������
���ݱ���ʹ���⣬��ȱ��һ��
��������1��д����ѧ����ʽ������п����������������������
��2�����������е���Ϣ�г���ϵʽ��
��3������Һ��˵���μӻ�ѧ��Ӧ�����ʵ�������ָ���ʵ�����������DZ�����Һ��һ��ˮ�ֱ����������ʾͻ�����������
��2�����������е���Ϣ�г���ϵʽ��
��3������Һ��˵���μӻ�ѧ��Ӧ�����ʵ�������ָ���ʵ�����������DZ�����Һ��һ��ˮ�ֱ����������ʾͻ�����������
����⣺��1�������ɵ�H2����ΪX��
Zn+H2SO4�TZnSO4+H2��
65 2
6.5�� X
�T
�ã�X=0.2��
��2�����������е���Ϣ��֪���ʴ�Ϊ��22.4L��0.2 g/2g��
��3������Һ��˵���μӻ�ѧ��Ӧ�����ʵ�������ָ���ʵ����������ȱ��������Һ�����ʵ���������������������������ΪY��
Zn+H2SO4�TZnSO4+H2��
98 2
245a Y
�T
�ã�Y=5a
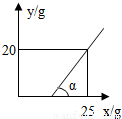
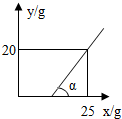
����DZ�����Һ��һ��ˮ�ֱ����������ʾͻ�������������ͼ����Կ������տ�ʼһ��ʱ�䣬��Ȼˮ�ֱ���������������û��������˵����Һ���DZ�����Һ��
�ߦ�=45�㣬��Ӧ��ֱ���������ǵ���ֱ�������Σ����ͼ����Կ���������20��ˮ�����������������20�ˣ�Ҳ����˵20�����ʺ�20��ˮ��ɵ���Һ���DZ�����Һ����˱�����Һ�����ʵ���������=
��100%�T50%��
�ʴ�Ϊ��������Һ������������5a�� �����ͣ� 50%��
Zn+H2SO4�TZnSO4+H2��
65 2
6.5�� X
| 65 |
| 2 |
| 6.5�� |
| X |
�ã�X=0.2��
��2�����������е���Ϣ��֪���ʴ�Ϊ��22.4L��0.2 g/2g��
��3������Һ��˵���μӻ�ѧ��Ӧ�����ʵ�������ָ���ʵ����������ȱ��������Һ�����ʵ���������������������������ΪY��
Zn+H2SO4�TZnSO4+H2��
98 2
245a Y
| 98 |
| 2 |
| 245a |
| Y |
�ã�Y=5a
����DZ�����Һ��һ��ˮ�ֱ����������ʾͻ�������������ͼ����Կ������տ�ʼһ��ʱ�䣬��Ȼˮ�ֱ���������������û��������˵����Һ���DZ�����Һ��
�ߦ�=45�㣬��Ӧ��ֱ���������ǵ���ֱ�������Σ����ͼ����Կ���������20��ˮ�����������������20�ˣ�Ҳ����˵20�����ʺ�20��ˮ��ɵ���Һ���DZ�����Һ����˱�����Һ�����ʵ���������=
| 20�� |
| 20��+20�� |
�ʴ�Ϊ��������Һ������������5a�� �����ͣ� 50%��
������ѧ�����ͼ��ķ�����һ�㿴ת�۵��ͼ������ƣ�ע�����ĸ�ʽ�淶�ԣ�

��ϰ��ϵ�д�
 ѧϰʵ����ϵ�д�
ѧϰʵ����ϵ�д�
�����Ŀ
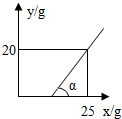 ��6.5��п��������ϡ���ᷴӦ��
��6.5��п��������ϡ���ᷴӦ��