��Ŀ����
��ѧ������������ϢϢ��أ�������ѧ֪ʶ�ش��������⣮
��1��ʳ���dz��õĵ�ζƷ�������ԣ�pH 7��
��2��������ũҵ�����г��õ�һ�ֻ��ʣ��仯ѧʽ�� �������� �ʣ���ʡ��ʡ��طʡ����Ϸʣ�
��3����Ȼ������Ҫ�ɷ��� ����ȼ�յĻ�ѧ����ʽ�ǣ�
��4����˾ƥ����һ�ֳ��õĽ�����ʹҩ���仯ѧʽ��C9H8O4������ Ԫ����ɣ���Է��������� ��C��H��O����Ԫ�ص�������Ϊ ��
��1��ʳ���dz��õĵ�ζƷ�������ԣ�pH
��2��������ũҵ�����г��õ�һ�ֻ��ʣ��仯ѧʽ��
��3����Ȼ������Ҫ�ɷ���
��4����˾ƥ����һ�ֳ��õĽ�����ʹҩ���仯ѧʽ��C9H8O4������
���㣺��Һ���������pHֵ�Ĺ�ϵ,�������ʵ����������,��ѧʽ����д������,Ԫ�������ȵļ���,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ,����ȼ�ϵ�ʹ������Ի�����Ӱ��
ר�⣺��ѧ����Դ,��ѧ����������غ㶨��,�������� ���ͨ��,�������� ��ѧ����
�������������е�֪ʶ���з�����������Һ��pHС��7���������صĻ�ѧʽ�Լ������������н����Ȼ������Ҫ�ɷ��Ǽ��飬��ȫȼ�����ɵ��Ƕ�����̼��ˮ�����ݰ�˾ƥ�ֵĻ�ѧʽ���н�ɣ�
����⣺��1��������Һ��pHС��7���������
��2�����صĻ�ѧʽΪCO��NH2��2�����е�Ӫ��Ԫ���ǵ�Ԫ�أ����ڵ��ʣ����CO��NH2��2������
��3����Ȼ������Ҫ�ɷ��Ǽ��飬��ȫȼ�����ɵ��Ƕ�����̼��ˮ��������飬CH4+2O2
CO2+2H2O��
��4�����ݰ�˾ƥ�ֵĻ�ѧʽC9H8O4�����Կ�����������̼���⡢������Ԫ����ɣ���Է���������12��9+1��8+16��4=180��C��H��O����Ԫ�ص�������Ϊ12��9��1��8��16��4=27��2��16���������180��27��2��16��
��2�����صĻ�ѧʽΪCO��NH2��2�����е�Ӫ��Ԫ���ǵ�Ԫ�أ����ڵ��ʣ����CO��NH2��2������
��3����Ȼ������Ҫ�ɷ��Ǽ��飬��ȫȼ�����ɵ��Ƕ�����̼��ˮ��������飬CH4+2O2
| ||
��4�����ݰ�˾ƥ�ֵĻ�ѧʽC9H8O4�����Կ�����������̼���⡢������Ԫ����ɣ���Է���������12��9+1��8+16��4=180��C��H��O����Ԫ�ص�������Ϊ12��9��1��8��16��4=27��2��16���������180��27��2��16��
���������⿼������Һ���������pH�Լ����ݻ�ѧʽ�ļ���ͻ�ѧ����ʽ����д��֪ʶ����ɴ��⣬�����������е�֪ʶ���У�

��ϰ��ϵ�д�
�����Ŀ
�ø����ʯ��ֽ��̽��������̼��ˮ��Ӧ����ʱ������ʵ�鲻��Ҫ̽�����ǣ�������
A�� ��ϡ���� |
B�� ��ˮ |
C�� ֱ�ӷ��������̼�� |
D�� ��ˮ����������̼�� |
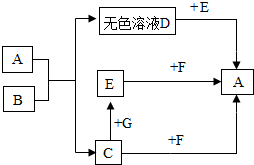 ͼ�е�A��B��C��D��E��F��G���dz��л�ѧ�еij������ʣ�����A��һ���Σ��������������ϣ�B������θҺ��������һ���ᣬC��ֲ�������õ�һ��ԭ�ϣ�E���Σ��㷺���ڲ�������ֽ����֯��ϴ�Ӽ��������ȣ�F��G�Ǽ����֮���������ת����ϵ��ͼ�в�������������ȥ����
ͼ�е�A��B��C��D��E��F��G���dz��л�ѧ�еij������ʣ�����A��һ���Σ��������������ϣ�B������θҺ��������һ���ᣬC��ֲ�������õ�һ��ԭ�ϣ�E���Σ��㷺���ڲ�������ֽ����֯��ϴ�Ӽ��������ȣ�F��G�Ǽ����֮���������ת����ϵ��ͼ�в�������������ȥ����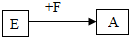 �Ļ�ѧ��Ӧ����ʽ��
�Ļ�ѧ��Ӧ����ʽ��