��Ŀ����
Ϊ�ⶨʵ������ijʯ��ʯ��Ʒ�Ĵ��ȣ���ȡ25gʯ��ʯ�����ʲ���̼Ԫ���Ҳ������ᷴӦ��������ϡ����ǡ����ȫ��Ӧ���ռ���8.8g������̼���塣
��1����ѧ��Ӧǰ��Ԫ�ص�������������䣬�ݴ˿�֪��ʯ��ʯ��Ʒ��̼Ԫ�ص������� ���г�����ʽ��
��2������Ҫ����������Ʒ�к�̼��Ƶ�������
����һ�����û�ѧʽ���м���
�����������û�ѧ����ʽ���м���
��3�������ʯ��ʯ��Ʒ�Ĵ��ȣ�����= ��
��
��1����ѧ��Ӧǰ��Ԫ�ص�������������䣬�ݴ˿�֪��ʯ��ʯ��Ʒ��̼Ԫ�ص������� ���г�����ʽ��
��2������Ҫ����������Ʒ�к�̼��Ƶ�������
����һ�����û�ѧʽ���м���
�����������û�ѧ����ʽ���м���
��3�������ʯ��ʯ��Ʒ�Ĵ��ȣ�����=
 ��
�� Ϊ�ⶨʵ������ijʯ��ʯ��Ʒ�Ĵ��ȣ���ȡ25gʯ��ʯ�����ʲ���̼Ԫ���Ҳ������ᷴӦ��������ϡ����ǡ����ȫ��Ӧ���ռ���8.8g������̼���塣
��1��8.8g �� 12��44��100%=2.4g
��2������һ�� 2.4g ��12��100 = 20g �����������ԡ�
��3��20g��25g ��100%=80%
��1��8.8g �� 12��44��100%=2.4g
��2������һ�� 2.4g ��12��100 = 20g �����������ԡ�
��3��20g��25g ��100%=80%

��ϰ��ϵ�д�
�����Ŀ
��2012?��ˮ�ض�ģ����ʯ���������ƺ��������ƵĹ��������ʵ���ҳ��õĸ�����������Ϣ���±���ʾ��ͬѧ��Ϊȷ��һƿ���õġ���ʯ�ҡ��������Ѳ��ֱ��ʻ�ȫ�����ʣ���Ʒ�ijɷ֣���������̽����
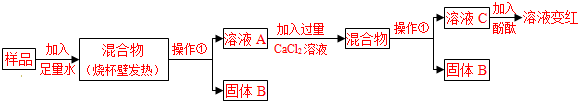
| [С����] �ټ�ʯ���������ơ��������ƵĹ�����������տ����е�ˮ�����Ͷ�����̼�� ���Ȼ�����Һ�����ԣ�[�������]ʵ�����о��õļ�ʯ����Ʒ�ijɷ���ʲô�� [���в���]���õļ�ʯ����Ʒ�п��ܺ���CaO��NaOH��Na2CO3��Ca��OH��2��CaCO3�ȳɷ֣��û�ѧ����ʽ��ʾ��Ʒ�к���Ca��OH��2��Na2CO3���ܺ��е�ԭ�� CaO+H2O�TCa��OH��2����2NaOH+CO2�TNa2CO3+H2O�ȣ��𰸺������ɣ� CaO+H2O�TCa��OH��2����2NaOH+CO2�TNa2CO3+H2O�ȣ��𰸺������ɣ� ��дһ������[̽������]  �Իش��������⣺ ��1�������ٵ������� ���� ���� ����ҺC��һ����OH- OH- ���ӣ���2����ҺA�м���CaCl2��Һ��������壬˵����Ʒ��һ������ ̼���� ̼���� ����Ӧ�ķ���ʽΪNa2CO3+CaCl2�T2NaCl+CaCO3�� Na2CO3+CaCl2�T2NaCl+CaCO3�� ����3������ҺA�м���CaCl2��Һ��֤��CaCl2��Һ�����ķ����� ȡ��ҺC���Թ��еμ�̼������Һ���а�ɫ�������ɣ�˵��CaCl2��Һ�ѹ��� ȡ��ҺC���Թ��еμ�̼������Һ���а�ɫ�������ɣ�˵��CaCl2��Һ�ѹ��� ��[ʵ�����] ��������ʵ��������̼������ۺ϶���Һ����ɷֵ�̽�������ж���Ʒ�ɷֵķ�����ȷ���� �ڢ� �ڢ� ������ţ�������Ʒ��һ����NaOH ����Ʒ��һ����Na2CO3 ����Ʒ�к�NaOH��CaO�е�һ�ֻ����� [��չ����] Ϊ�ⶨʵ������һƿ���ʵ��ռ���NaOH�ĺ�����ijͬѧȡ�������ռ���Ʒ������һ������ˮ�õ�200g��Һ���ټ���200gϡ���ᣨ��������ֽ��赽���ٷų�����Ϊֹ��������ҺΪ395.6g����������㣺 ��1����Ӧ������CO2������Ϊ 4.4 4.4 g����2������ȡ�ռ���ƷΪ50.0g������Ʒ��NaOH�����������Ƕ��٣���д��������̣� 78.8%�� 78.8%�� ����3����ͬŨ�ȵ����ᣬ�ֱ���δ���ʡ����ֱ��ʡ���ȫ�����ʣ����ʶ���Na2CO3�����ռ���Ʒ��Ӧ����Ҫ�������������ȣ�����Ԫ�������غ�ĽǶȽ�����ԭ���ǣ� �������ƺ�̼���ƶ���ÿ46g��Ԫ������142g�����ƣ�����98g���� �������ƺ�̼���ƶ���ÿ46g��Ԫ������142g�����ƣ�����98g���� ��
��ʯ���������ƺ��������ƵĹ��������ʵ���ҳ��õĸ�����������Ϣ���±���ʾ��ͬѧ��Ϊȷ��һƿ���õġ���ʯ�ҡ��������Ѳ��ֱ��ʻ�ȫ�����ʣ���Ʒ�ijɷ֣���������̽����
[���в���]���õļ�ʯ����Ʒ�п��ܺ���CaO��NaOH��Na2CO3��Ca��OH��2��CaCO3�ȳɷ֣��û�ѧ����ʽ��ʾ��Ʒ�к���Ca��OH��2��Na2CO3���ܺ��е�ԭ��______��дһ������ [̽������]  �Իش��������⣺ ��1�������ٵ�������______����ҺC��һ����______���ӣ� ��2����ҺA�м���CaCl2��Һ��������壬˵����Ʒ��һ������______����Ӧ�ķ���ʽΪ______�� ��3������ҺA�м���CaCl2��Һ��֤��CaCl2��Һ�����ķ�����______�� [ʵ�����] ��������ʵ��������̼������ۺ϶���Һ����ɷֵ�̽�������ж���Ʒ�ɷֵķ�����ȷ����______������ţ��� ����Ʒ��һ����NaOH ����Ʒ��һ����Na2CO3 ����Ʒ�к�NaOH��CaO�е�һ�ֻ����� [��չ����] Ϊ�ⶨʵ������һƿ���ʵ��ռ���NaOH�ĺ�����ijͬѧȡ�������ռ���Ʒ������һ������ˮ�õ�200g��Һ���ټ���200gϡ���ᣨ��������ֽ��赽���ٷų�����Ϊֹ��������ҺΪ395.6g����������㣺 ��1����Ӧ������CO2������Ϊ______ g�� ��2������ȡ�ռ���ƷΪ50.0g������Ʒ��NaOH�����������Ƕ��٣���д��������̣�______�� ��3����ͬŨ�ȵ����ᣬ�ֱ���δ���ʡ����ֱ��ʡ���ȫ�����ʣ����ʶ���Na2CO3�����ռ���Ʒ��Ӧ����Ҫ�������������ȣ�����Ԫ�������غ�ĽǶȽ�����ԭ���ǣ�______�� |
 ��ʯ����ʵ���ҳ��õĸ������ͬѧ��Ϊȷ��һƿ���õġ���ʯ�ҡ��������Ѳ��ֱ��ʻ�ȫ�����ʣ���Ʒ�ijɷ֣���������̽����
��ʯ����ʵ���ҳ��õĸ������ͬѧ��Ϊȷ��һƿ���õġ���ʯ�ҡ��������Ѳ��ֱ��ʻ�ȫ�����ʣ���Ʒ�ijɷ֣���������̽����