��Ŀ����
����Ŀ��̼��̼�Ļ������ڹ�ũҵ�������ճ��������й㷺����;��
��1����ͼ��̼ԭ�ӽṹʾ��ͼ![]() ��̼ԭ�ӵ���������________________��̼Ԫ����Ԫ�����ڱ���λ�ڵ�__________���ڡ�
��̼ԭ�ӵ���������________________��̼Ԫ����Ԫ�����ڱ���λ�ڵ�__________���ڡ�
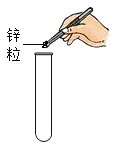
��2���Ŵ���ī��д����Ƶ��ֻ�������Բ���ɫ��ԭ����ī����Ҫ�ɷ�̼�ڳ����¾���___________�ԡ�
��3��������̼�ܲ�����������ɴ����еġ�̼ѭ���������Ǵ����еĶ�����̼�ĺ���������������ʹȫ���ů���Ӷ�����_____________���߲ˡ��������������ʡ��Ķ�����̼�����ö��ַ����Ƶã����������ϡ���ᣨH2SO4����̼����泥�NH4HCO3����Ӧ�Ƶã���Ӧ����������泥�ˮ�Ͷ�����̼���÷�Ӧ�Ļ�ѧ����ʽΪ______________________
���𰸡���1��6��������2���ȶ�����3������ЧӦ��H2SO4+2NH4HCO3=(NH4)2SO4+2H2O+2CO2��
��������
���������̼ԭ�ӵ���������6��̼Ԫ����Ԫ�����ڱ���λ�ڵ�2���ڣ���Ϊһ�����������Ӳ㣻�Ŵ���ī��д����Ƶ��ֻ�������Բ���ɫ��ԭ����ī����Ҫ�ɷ�̼�ڳ����¾����ȶ��ԣ�������̼�ܲ�����������ɴ����еġ�̼ѭ���������Ǵ����еĶ�����̼�ĺ���������������ʹȫ���ů���Ӷ���������ЧӦ�߲ˡ��������������ʡ��Ķ�����̼�����ö��ַ����Ƶã����������ϡ���ᣨH2SO4����̼����泥�NH4HCO3����Ӧ�Ƶã���Ӧ����������泥�ˮ�Ͷ�����̼����Ӧ�Ļ�ѧ����ʽΪH2SO4+2NH4HCO3=(NH4)2SO4+2H2O+2CO2����

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�����Ŀ�����������еij���Ԫ�أ�ȱ��ʱ��ͨ��ʳ�ñ���ҩ����������������ij����ҩ����Ҫ�ɷ�Ϊ̼��ƣ��ֽ�100g����ֳ�5�ȷݣ���μӵ���40g��ҩ���Ƴɵķ�ĩ��(�����ɷֲ������ᷴӦ)���õ�����������ͼ��������й���Ϣ�ش����⣺
��� | ��1�� | ��2�� | ��3�� | ��4�� | ��5�� |
�������������/g | 20 | 20 | 20 | 20 | 20 |
ʣ����������/g | 35 | 30 | 25 | 20 | a |

(1)a����ֵΪ___����Ʒ�Ʋ���ҩ����CaCO3������������___��
(2)������������ʵ���������____��(д��������̣�����������һλС��)
(3)��������Ƹ�����ʱ��������ƫС�ˣ��Ը�Ʒ�Ʋ���ҩ����CaCO3���������������Ƿ���Ӱ�졣��Ŀ�����___��
����Ŀ���Ȼ��ƺ�̼���ƾ�����ܽ��(0~30 ��C) ���±���ʾ:
�¶� | 0��C | l0 ��C | 20 ��C | 30 ��C |
NaCl(g) | 35.7 | 35.8 | 36.0 | 36.3 |
Na2CO3����(g) | 7.0 | 12.5 | 21.5 | 38.8 |
��1�������ϱ�����,̼���ƾ�����ܽ�����¶�____________________________
��2������ˮ��100g���Ȼ�����̼���Ƶı�����Һ����30��C����0��Cʱ�������Ȼ��ƾ��������____________��(������������С��������������)̼���ƾ����������
��3�����Ӻ�������NaCl���ʵ�̼����Ũ��Һ�з����̼���ƾ���,Ӧ��ȡ____________������
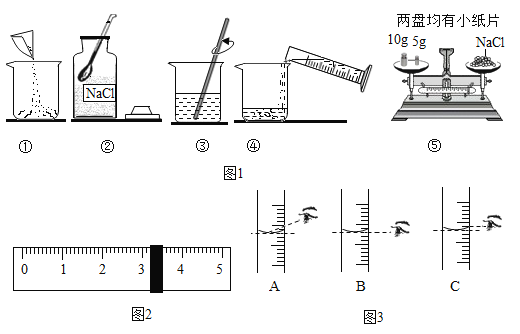
��4��20��Cʱ��NaCl�ܽ���ˮ��ʵ���������±���������������ȷ����____________��
ʵ����� | ˮ������(g) | ����NaCl����(g) | ��Һ������(g) |
�� | 10 | 2 | 12 |
�� | 10 | 3 | 13 |
�� | 10 | 4 | 13.6 |
�� | 10 | 5 | 13.6 |
A ��������Һ��������������Ϊ20%
B ����������Һ�DZ�����Һ
C 20��Cʱ10gˮ������ܽ�4gNaCl
D �ۢ���Һ�����������������