��Ŀ����
�����ǵ���Ҫ�ɷ���̼��ƣ�Ϊ�˲ⶨ������̼��Ƶ�����������С�պ�С��ͬѧ����������ʵ�飮��ش�������⣺
��1������������ϴ�������ﲢ��������ձ��Ȼ�����ձ��м����������ᣬ�ڵ����Ϻܿ��������С���ݲ��������ռ���������ͨ�����ʯ��ˮ�У�ʯ��ˮ����ǣ��ݴ˿��жϣ��ռ����������к���______��
��2��ʵ����̺Ͳⶨ�����ʵ������������ʾ��
С�ո��ݡ����Dz�������Ϊ4.3g������õ�����̼��Ƶ���������Ϊ______��С�¸��ݡ���Ӧ�����м��ٵ���������Ҳ����˵�����̼��Ƶ�����������������������������������������д��С�µļ�����̺ͽ����
��3��С�պ�С�¸��ݲ�ͬ��������õ�����̼��Ƶ��������������Բ������������п��ܵ�ԭ��
�⣺��1�����ݶ�����̼�����ʿ���֪����������̼�ܹ�ʹ����ʯ��ˮ����ǣ����Կ����жϸù����в����˶�����̼��
��2����̼������ᷴӦ�����������Ϊ4.3g��̼��Ƶ�����Ϊ10g-4.3g=5.7g��
����̼��Ƶ���������Ϊ��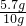 ��100%=57%��
��100%=57%��
С�µļ������Ϊ��CO2 ������Ϊ��10g+100g-106.7g=3.3g
��̼��Ƶ�����Ϊx���Ȼ��������Ϊy
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 73 44
x y 3.3g
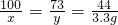
��ã�x=7.5g�� y=5.5g��
ͨ������ã�̼��Ƶ���������Ϊ��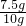 ��100%=75%��
��100%=75%��
������������������� ��100%=5.5%��
��100%=5.5%��
�𣺵�����̼��Ƶ���������Ϊ75%��
��3��С�ռ������õĵ��Dz�������δ���ʵ�ʵ��Dz�������С��4.3g��
��С�¼������õ����ݡ�3.3g���а����˷�Ӧ����������ӷ��˵��Ȼ����������������ʵ�����ɵĶ�����̼��������С��3.3g��
��С�պ�С�¸��ݲ�ͬ��������õ�����̼��Ƶ��������������Բ���ԭ�����Ϊ��������δ����ͳ��������岻������ɵģ�
�ʴ�Ϊ����1��������̼��
��2��57%��75%��5.5%��
��3����������δ����ͳ��������岻������ɵģ�
��������1�����ݶ�����̼�����ʿ���֪����������̼�ܹ�ʹ����ʯ��ˮ����ǣ����Ծݴ˽��
��2�����ݼ����ǵ���Ҫ�ɷ���̼��ƣ����ᷴӦ�Dz�������Ϊ4.3g����̼��������ᷴӦ����ɼ���̼��Ƶ��������ټ��㵰����̼��Ƶ�����������
��3������ʵ���й��������ij�����������ռ��ȷ�������������
���������⿼��ѧ�����û�ѧ��Ӧ����ʽ�ļ��㣬��ȷ��Ӧ����������������������غ������������̼�������ǽ��Ĺؼ���
��2����̼������ᷴӦ�����������Ϊ4.3g��̼��Ƶ�����Ϊ10g-4.3g=5.7g��
����̼��Ƶ���������Ϊ��
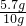 ��100%=57%��
��100%=57%��С�µļ������Ϊ��CO2 ������Ϊ��10g+100g-106.7g=3.3g
��̼��Ƶ�����Ϊx���Ȼ��������Ϊy
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 73 44
x y 3.3g
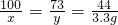
��ã�x=7.5g�� y=5.5g��
ͨ������ã�̼��Ƶ���������Ϊ��
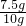 ��100%=75%��
��100%=75%���������������������
 ��100%=5.5%��
��100%=5.5%���𣺵�����̼��Ƶ���������Ϊ75%��
��3��С�ռ������õĵ��Dz�������δ���ʵ�ʵ��Dz�������С��4.3g��
��С�¼������õ����ݡ�3.3g���а����˷�Ӧ����������ӷ��˵��Ȼ����������������ʵ�����ɵĶ�����̼��������С��3.3g��
��С�պ�С�¸��ݲ�ͬ��������õ�����̼��Ƶ��������������Բ���ԭ�����Ϊ��������δ����ͳ��������岻������ɵģ�
�ʴ�Ϊ����1��������̼��
��2��57%��75%��5.5%��
��3����������δ����ͳ��������岻������ɵģ�
��������1�����ݶ�����̼�����ʿ���֪����������̼�ܹ�ʹ����ʯ��ˮ����ǣ����Ծݴ˽��
��2�����ݼ����ǵ���Ҫ�ɷ���̼��ƣ����ᷴӦ�Dz�������Ϊ4.3g����̼��������ᷴӦ����ɼ���̼��Ƶ��������ټ��㵰����̼��Ƶ�����������
��3������ʵ���й��������ij�����������ռ��ȷ�������������
���������⿼��ѧ�����û�ѧ��Ӧ����ʽ�ļ��㣬��ȷ��Ӧ����������������������غ������������̼�������ǽ��Ĺؼ���

��ϰ��ϵ�д�
�����Ŀ