��Ŀ����
�ݱ�������2005��1��1���𣬹���ʡ���ػ������Ž���ʵ��������Ⱦʵʩ�ϸ�Ļ�����ܣ�����ѧУ��ѧʵ����Ҫ�ŷųɷָ��ӵ���Ⱦ�����Ҳ����Ϊ������ܷ�Χ��ijУ��ѧ��ȤС���ͬѧ�ڼ�ʵ�����н����������ֱ���̼��������Ӧ��ʵ���Ϊ�˽��ʵ�����������Կ����ɷ���ɵ�Ӱ�죬�������������ʵ��װ�ý���ʵ�飨ͼ�ж�����ݵ������ǣ�������������Һ�ĽӴ������ʹ��Ӧ��ֽ��У���
��ش��������⣺
��1��ʵ����û������Ⱦ����ʵ�����н��У�ȡ��������ʵ�����п�����Ʒ�ķ�����______��
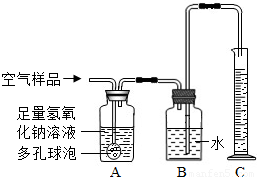
��2����ȡ�õĿ�����Ʒ����ͼ��ʾ��װ�ý���ʵ��ⶨ��װ��A��������______�����з�����Ӧ�Ļ�ѧ����ʽΪ______װ��B�е�������______��װ��C��������______��
��3����ͨ�������Ʒ100mL��ʵ���������Ͳ��Һ������Ϊ99mL��������Һ����Բ��ƣ���˵��װ��______��������1mL���壮����Щ��������ŷŵ������У������______����Ի�����ɲ���Ӱ�죮
��4����д����������ѧʵ��ʱ������ʵ��Ի�����Ⱦ��һ��������______��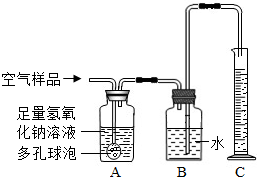
��ش��������⣺
��1��ʵ����û������Ⱦ����ʵ�����н��У�ȡ��������ʵ�����п�����Ʒ�ķ�����______��
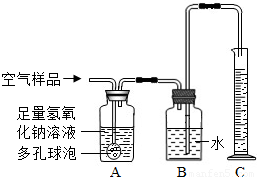
��2����ȡ�õĿ�����Ʒ����ͼ��ʾ��װ�ý���ʵ��ⶨ��װ��A��������______�����з�����Ӧ�Ļ�ѧ����ʽΪ______װ��B�е�������______��װ��C��������______��
��3����ͨ�������Ʒ100mL��ʵ���������Ͳ��Һ������Ϊ99mL��������Һ����Բ��ƣ���˵��װ��______��������1mL���壮����Щ��������ŷŵ������У������______����Ի�����ɲ���Ӱ�죮
��4����д����������ѧʵ��ʱ������ʵ��Ի�����Ⱦ��һ��������______��

��1��ȡ��������ʵ�����п�����Ʒ�ķ����У�������ƿװ��ˮ��ë����Ƭ�Ǻã������ʵ���ҽ�ˮ�������ٸ���ë����Ƭ���ɣ�Ҳ������ע������ȡ��
��2�������ڼ�ʵ�����н����������ֱ���̼��������Ӧ��ʵ�飬�����ɶ�����̼�������������壬�������������嶼�����������壬����Ӧ������Ϊ����������Һ���ڼ������������巴Ӧ������װ��A������������������Ʒ�еĶ�����̼�Ͷ�������ȥ���������ʣ�����������Bװ�ã�ʹ��ѹǿ����Һ��ѹ��C�У�����C��ˮ������Ƕ��٣���ô��B �е�����������Ƕ��٣�
��3����ͨ�������Ʒ100mL��ʵ���������Ͳ��Һ������Ϊ99mL�����Ա����յ�����������1mL����Ϊ���������Ƕ�����̼��������������̼���˻��������ЧӦ���������������������ꣻ
��4������ͬѧ����ʵ�����л����ܴ�����Ⱦ�����壬����ֻ��������ѧʵ��ʱ������ʵ��ҩƷ�����ٶԻ�����Ⱦ�������ŷţ��ʴ�Ϊ����1����ע������ȡ�����ü���ƿװ��ˮ�������ڽ�ˮ���������ɣ�ƿ�����ϣ����ϲ���Ƭ��ʹ�������ռ��ȣ�
��2������������Ʒ�еĶ�����̼�Ͷ�������2NaOH+CO2=Na2CO3+H2O��2NaOH+SO2=Na2SO3+H2O��ˮ���ŵ��ܽ���C�У���ˮλ�½���������������̼�Ͷ����������պ�ʣ������Ĵ������
��3��A�����������ЧӦ
��4������ʵ��ҩƷ�����������Ⱦ����д��������ŷš����ҵ���Һ�����ӷ���ȣ�
��2�������ڼ�ʵ�����н����������ֱ���̼��������Ӧ��ʵ�飬�����ɶ�����̼�������������壬�������������嶼�����������壬����Ӧ������Ϊ����������Һ���ڼ������������巴Ӧ������װ��A������������������Ʒ�еĶ�����̼�Ͷ�������ȥ���������ʣ�����������Bװ�ã�ʹ��ѹǿ����Һ��ѹ��C�У�����C��ˮ������Ƕ��٣���ô��B �е�����������Ƕ��٣�
��3����ͨ�������Ʒ100mL��ʵ���������Ͳ��Һ������Ϊ99mL�����Ա����յ�����������1mL����Ϊ���������Ƕ�����̼��������������̼���˻��������ЧӦ���������������������ꣻ
��4������ͬѧ����ʵ�����л����ܴ�����Ⱦ�����壬����ֻ��������ѧʵ��ʱ������ʵ��ҩƷ�����ٶԻ�����Ⱦ�������ŷţ��ʴ�Ϊ����1����ע������ȡ�����ü���ƿװ��ˮ�������ڽ�ˮ���������ɣ�ƿ�����ϣ����ϲ���Ƭ��ʹ�������ռ��ȣ�
��2������������Ʒ�еĶ�����̼�Ͷ�������2NaOH+CO2=Na2CO3+H2O��2NaOH+SO2=Na2SO3+H2O��ˮ���ŵ��ܽ���C�У���ˮλ�½���������������̼�Ͷ����������պ�ʣ������Ĵ������
��3��A�����������ЧӦ
��4������ʵ��ҩƷ�����������Ⱦ����д��������ŷš����ҵ���Һ�����ӷ���ȣ�

��ϰ��ϵ�д�
 ���Ͱ�ͨ�������Сѧ��ʱͬ�����ϵ�д�
���Ͱ�ͨ�������Сѧ��ʱͬ�����ϵ�д� ���Ͱ�ͨ������ϵ�д�
���Ͱ�ͨ������ϵ�д� �ٷ�ѧ����ҵ��������ϵ�д�
�ٷ�ѧ����ҵ��������ϵ�д�
�����Ŀ
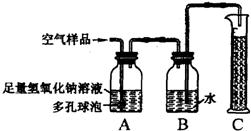 22���ݱ�������2005��1��1���𣬹���ʡ���ػ������Ž���ʵ��������Ⱦʵʩ�ϸ�Ļ�����ܣ�����ѧУ��ѧʵ����Ҫ�ŷųɷָ��ӵ���Ⱦ�����Ҳ����Ϊ������ܷ�Χ��ijУ��ѧ��ȤС���ͬѧ�ڼ�ʵ�����н����������ֱ���̼��������Ӧ��ʵ���Ϊ�˽��ʵ�����������Կ����ɷ���ɵ�Ӱ�죬�������������ʵ��װ�ý���ʵ�飨ͼ�ж�����ݵ������ǣ�������������Һ�ĽӴ������ʹ��Ӧ��ֽ��У���
22���ݱ�������2005��1��1���𣬹���ʡ���ػ������Ž���ʵ��������Ⱦʵʩ�ϸ�Ļ�����ܣ�����ѧУ��ѧʵ����Ҫ�ŷųɷָ��ӵ���Ⱦ�����Ҳ����Ϊ������ܷ�Χ��ijУ��ѧ��ȤС���ͬѧ�ڼ�ʵ�����н����������ֱ���̼��������Ӧ��ʵ���Ϊ�˽��ʵ�����������Կ����ɷ���ɵ�Ӱ�죬�������������ʵ��װ�ý���ʵ�飨ͼ�ж�����ݵ������ǣ�������������Һ�ĽӴ������ʹ��Ӧ��ֽ��У���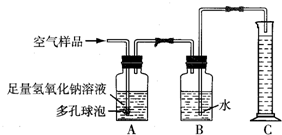 A��ij���������᳧���豸��ª�������¾ɣ��ó�ÿ���ŷŴ�����SO2�ķ����ͺ�H2SO4�����Է�ˮ�����ص����������;������ú̿��ȼ�ϣ�ֻҪ����������꣬�Ը�����ɼ����ƻ���
A��ij���������᳧���豸��ª�������¾ɣ��ó�ÿ���ŷŴ�����SO2�ķ����ͺ�H2SO4�����Է�ˮ�����ص����������;������ú̿��ȼ�ϣ�ֻҪ����������꣬�Ը�����ɼ����ƻ���