��Ŀ����
��2008?������һģ����ͼ��ʵ���ҳ��õ�ʵ����������Ҫ������������⣮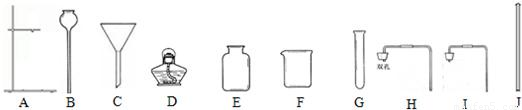
��1����д��������������ƣ�C______J______
��2�������ᴿʵ���У��ܽ���ε�����������______��
��ʵ��ǰ�����ۣ����ʵ���������������������������ȷ����______��
A���������ƾ����������������ϵľƾ�ȼ��������������ˮ����
B�����˻��ǵ�Һ��ʱ������̫�����ò���������
C�������������ֽ϶���ʳ�ι���ʱ��Ӧֹͣ����
����ϣ���ܽϿ���ܽ�ʳ�Σ���Ľ��鼰�����ǣ�______
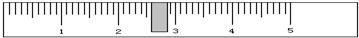
����������ƽ�������õ��ľ��Σ���5�˵��������______������ҡ����̣���ƽƽ��ʱ����������ͼ��ʾ�����ε�����Ϊ______�ˣ�
��3����������������װʵ������ȡ�����װ�ã�
��ʵ�����Ʊ�������̼�ķ�Ӧ����ʽΪ______������װ��Ӧѡ�õ�������______������ţ���
���ø��������ԭ���Ʊ������ķ�Ӧ����ʽ______ K2MnO4+MnO2+O2��
���𰸡���������1����Ϥ�����������˽����ƣ�
��2�����ܽ����ʵ�������һ�����ձ���
���ܽ��ٶȵ�������Ҫ�漰��������أ��¶ȡ����衢��ϸ�Dz��õ���Ҫ��ʽ��
�۸��ݡ��������롱��ԭ���жϣ�������������=��������+�����������е�ʽ����������ʵ��������
��3�������ʵ������ȡ������̼�ķ���ʽ�����ݷ�Ӧ���״̬�ͷ�Ӧ����ѡ��������
����Ǹ��������ȡ�����Ļ�ѧ��Ӧʽ�����ݷ�Ӧ���״̬�ͷ�Ӧ����ѡ��������
����⣺��1��ͼ��C��©����J�Dz�������
�ʴ�Ϊ��©������������
��2���ܽ����ʵ�������һ�����ձ���
�ʴ�Ϊ���ձ���
���������ϵľƾ�ȼ��������Ӧ��Ĩ�����𣻹���ʵ��ʱ�������ò��������裻�����������ֽ϶���ʳ�ι���ʱ��Ӧֹͣ���ȣ���������������Һ�壮
��ѡC��
���������ܽ���ٶȿɲ��÷�����Ҫ��1������ܼ��¶ȣ�ˮ�£���2�����裻3�����������Ū��С�ŵĵȷ�ʽ��
�ʴ�Ϊ���ò�����������ι����������ȣ�
������������=��������+�����������е�ʽ�����ε�����=5g+2.6g=7.6g��
�ʴ�Ϊ���ң�7.6g��
��3����ʵ�����ô���ʯ��ϡ���ᷴӦ��ȡ������̼�Ļ�ѧ��Ӧʽ�ǣ�CaCO3+2HCl�TCaCl2+H2O+CO2������˷�Ӧ�ǡ���Һ�����ȡ��ͷ�Ӧ�����Է���װ�ÿ�ѡ�� AG��I����ABG��H����
�ʴ�Ϊ��CaCO3+2HCl�TCaCl2+H2O+CO2����AG��I����ABG��H����
���ø��������ȡ�����Ļ�ѧ��Ӧʽ�ǣ�2KMnO4 K2MnO4+MnO2+O2�����˷�Ӧ�ǹ���ļ��ȷ�Ӧ����ѡȡ�ķ���װ���ǣ�ADG��
K2MnO4+MnO2+O2�����˷�Ӧ�ǹ���ļ��ȷ�Ӧ����ѡȡ�ķ���װ���ǣ�ADG��
�ʴ�Ϊ��2KMnO4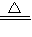 K2MnO4+MnO2+O2����ADG��
K2MnO4+MnO2+O2����ADG��
���������⿼���ܽ�ʵ�����õ�����������ʵ������е�ע�����������ƽ��ʹ�á�ʵ������ȡ�����Ͷ�����̼�Ļ�ѧ��Ӧʽ����д������������֪ʶ�㿼��ȫ�棬����࣬�����ǶԻ���֪ʶ�Ŀ��飬�Ƿ�������Ҫ��ͬѧ�ǵ�֪ʶ������ϸ�ij̶ȣ�
��2�����ܽ����ʵ�������һ�����ձ���
���ܽ��ٶȵ�������Ҫ�漰��������أ��¶ȡ����衢��ϸ�Dz��õ���Ҫ��ʽ��
�۸��ݡ��������롱��ԭ���жϣ�������������=��������+�����������е�ʽ����������ʵ��������
��3�������ʵ������ȡ������̼�ķ���ʽ�����ݷ�Ӧ���״̬�ͷ�Ӧ����ѡ��������
����Ǹ��������ȡ�����Ļ�ѧ��Ӧʽ�����ݷ�Ӧ���״̬�ͷ�Ӧ����ѡ��������
����⣺��1��ͼ��C��©����J�Dz�������
�ʴ�Ϊ��©������������
��2���ܽ����ʵ�������һ�����ձ���
�ʴ�Ϊ���ձ���
���������ϵľƾ�ȼ��������Ӧ��Ĩ�����𣻹���ʵ��ʱ�������ò��������裻�����������ֽ϶���ʳ�ι���ʱ��Ӧֹͣ���ȣ���������������Һ�壮
��ѡC��
���������ܽ���ٶȿɲ��÷�����Ҫ��1������ܼ��¶ȣ�ˮ�£���2�����裻3�����������Ū��С�ŵĵȷ�ʽ��
�ʴ�Ϊ���ò�����������ι����������ȣ�
������������=��������+�����������е�ʽ�����ε�����=5g+2.6g=7.6g��
�ʴ�Ϊ���ң�7.6g��
��3����ʵ�����ô���ʯ��ϡ���ᷴӦ��ȡ������̼�Ļ�ѧ��Ӧʽ�ǣ�CaCO3+2HCl�TCaCl2+H2O+CO2������˷�Ӧ�ǡ���Һ�����ȡ��ͷ�Ӧ�����Է���װ�ÿ�ѡ�� AG��I����ABG��H����
�ʴ�Ϊ��CaCO3+2HCl�TCaCl2+H2O+CO2����AG��I����ABG��H����
���ø��������ȡ�����Ļ�ѧ��Ӧʽ�ǣ�2KMnO4
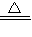 K2MnO4+MnO2+O2�����˷�Ӧ�ǹ���ļ��ȷ�Ӧ����ѡȡ�ķ���װ���ǣ�ADG��
K2MnO4+MnO2+O2�����˷�Ӧ�ǹ���ļ��ȷ�Ӧ����ѡȡ�ķ���װ���ǣ�ADG���ʴ�Ϊ��2KMnO4
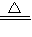 K2MnO4+MnO2+O2����ADG��
K2MnO4+MnO2+O2����ADG�����������⿼���ܽ�ʵ�����õ�����������ʵ������е�ע�����������ƽ��ʹ�á�ʵ������ȡ�����Ͷ�����̼�Ļ�ѧ��Ӧʽ����д������������֪ʶ�㿼��ȫ�棬����࣬�����ǶԻ���֪ʶ�Ŀ��飬�Ƿ�������Ҫ��ͬѧ�ǵ�֪ʶ������ϸ�ij̶ȣ�

��ϰ��ϵ�д�
�����Ŀ
��2008?������һģ���±���Ԫ�����ڱ���һ����
��1��ԭ������Ϊ14��Ԫ������Ϊ�����ԭ������Ϊ14��Ԫ�ط�����
��2��ÿ���ԭ���е��������ǣ���һ������ӵĺ����������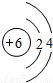
��3����ijԪ�ص�ԭ�ӽṹʾ��ͼ����Ԫ�����ϱ��е�λ���ǣ���ٻ�ڻ�ۣ���
��4�����Тڿո�Ԫ��Ӧ�����ڣ�ѡ��������ǽ����������䵥�ʵĻ�ѧ��Ա�����ѡ�ǿ��������������Ԫ���γɵ�����������Ϊ��ѡ�AO����A2O����OA����A2O3����
| �� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | |
| �ڶ����� | 3 Li � 7 | 4 Be �� 9 | 5 B �� 11 | �� | 7 N �� 14 | 8 O �� 16 | 9 F �� 19 | 10 Ne �� 20 |
| �������� | 11 Na �� 23 | �� | 13 Al �� 27 | 14 Si �� 28 | �� | 16 S �� 32 | 17 Cl �� 35.5 | 18 Ar � 40 |
��2��ÿ���ԭ���е��������ǣ���һ������ӵĺ����������
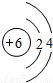
��3����ijԪ�ص�ԭ�ӽṹʾ��ͼ����Ԫ�����ϱ��е�λ���ǣ���ٻ�ڻ�ۣ���
��4�����Тڿո�Ԫ��Ӧ�����ڣ�ѡ��������ǽ����������䵥�ʵĻ�ѧ��Ա�����ѡ�ǿ��������������Ԫ���γɵ�����������Ϊ��ѡ�AO����A2O����OA����A2O3����
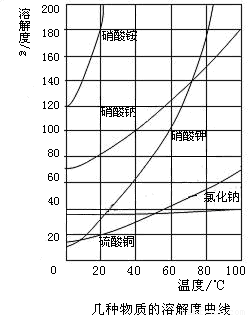
 ��2008?������һģ���۲���ͼ��ǩ���ش���������
��2008?������һģ���۲���ͼ��ǩ���ش���������