��Ŀ����
��2012?������(6��)��ҵ���á�̼����������CO��CO2��������CO2��������CO���������������ͼ��ʾ����������������ʾδ�������
(1)��Ӧ�ڷ������CO2���Ƴɸɱ����ɱ������� ��
(2)��Ӧ���ڡ���Ӧ�����ҡ��ڽ��С���NaOH����CaCO3�����IJ����� ��
(3)�����ҡ��з�����Ӧ�Ļ�ѧ����ʽΪ ��
(4)�����йظò����̵�������ȷ���� ��
| A��������CO2���Ʊ�����������Ʒ�����������������ŷ� |
| B������Ӧ�����ҡ��еķ�ӦҪ���մ����� |
| C�����������У�ֻ��һ�����ʿ�ѭ������ |
| D���ܺĴ��Ǹò�������һ��ȱ�� |
��1��������������2�����ˣ���3��CO2+2NaOH=Na2CO3+H2O����4��AD
����

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
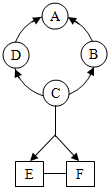 ��2012?��������ͼ��ʾA-F�dz��л�ѧ���������ʣ�ͼ�С�������ʾת����ϵ����-����ʾ��ܷ�Ӧ���������ʺͷ�Ӧ����δ�����������A���Ϻ�ɫ������B�����������壬C������θҺ�к��е��ᣮ
��2012?��������ͼ��ʾA-F�dz��л�ѧ���������ʣ�ͼ�С�������ʾת����ϵ����-����ʾ��ܷ�Ӧ���������ʺͷ�Ӧ����δ�����������A���Ϻ�ɫ������B�����������壬C������θҺ�к��е��ᣮ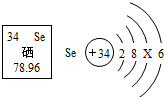 ��2012?����������Ӫ���ḻ����������ı������ܣ������и������ۡ������ʡ�ά����A��ά����B��ά����C��п�������ơ����ȣ�
��2012?����������Ӫ���ḻ����������ı������ܣ������и������ۡ������ʡ�ά����A��ά����B��ά����C��п�������ơ����ȣ�