��Ŀ����
22����1����pH��ֽ�ⶨ��Һ��pHʱ����ȷ�IJ�����
��2��С��ͬѧ��pH��ֽ������ȷ�����ⶨϴ�����ͻ�������pH�����ϴ������pH�Դ���7����ϴ������
��3����֪������������ͷ���Ľ�������ϴ��ʱ��Ӧ������
�ò�����մȡ����Һ�ڸ����pH��ֽ�ϣ������ɫ���Աȣ��ó�pHֵ
����2��С��ͬѧ��pH��ֽ������ȷ�����ⶨϴ�����ͻ�������pH�����ϴ������pH�Դ���7����ϴ������
��
�ԣ���û�������pH��С��7����������
�ԣ���3����֪������������ͷ���Ľ�������ϴ��ʱ��Ӧ������
ϴ��
������������
������������1��������PH��ֽ�ⶨ��ҺPHֵʱ�IJ���Ҫ�����ɣ�
��2������PH ����Һ����Թ�ϵ���ɣ�
��3�����ݣ�2�������������������ɣ�
��2������PH ����Һ����Թ�ϵ���ɣ�
��3�����ݣ�2�������������������ɣ�
����⣺��1����PH��ֽ�ⶨ��ҺPHֵʱ��ȷ�IJ����ǣ��ò�����մȡ����Һ�ڸ����pH��ֽ�ϣ������ɫ���Աȣ��ó�pHֵ��
��2��PH��7����Һ�����ԣ�PH=7��Һ�����ԣ�PH��7��Һ�Լ��ԣ�
�������⣺ϴ������pH�Դ���7�����Լ��ԣ���������pH��С��7���������ԣ�
�ʴ�Ϊ����
��3����Ϊ����������ͷ���Ľ���������ϴ��ʱ��Ӧ����ϴ���������û���������֤ͷ���������Ի����У�
�ʴ�Ϊ��ϴ��������������
��2��PH��7����Һ�����ԣ�PH=7��Һ�����ԣ�PH��7��Һ�Լ��ԣ�
�������⣺ϴ������pH�Դ���7�����Լ��ԣ���������pH��С��7���������ԣ�
�ʴ�Ϊ����
��3����Ϊ����������ͷ���Ľ���������ϴ��ʱ��Ӧ����ϴ���������û���������֤ͷ���������Ի����У�
�ʴ�Ϊ��ϴ��������������
��������PH��ֽ�ⶨ��ҺPHֵʱ��ȷ�IJ����ǣ��ò�����մȡ����Һ�ڸ����pH��ֽ�ϣ������ɫ���Աȣ��ó�pHֵ���мDz���ʪ��PH��ֽ��Ҳ���ܰ�PH��ֽ�ŵ�����Һ�У��Է���Ⱦ�Լ���

��ϰ��ϵ�д�
 ��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�
��Ǭ����������ҵ���ּ����ӱ����������ϵ�д�
�����Ŀ
ij��ȤС��ͬѧΪ֤��NaOH��Һ��ϡ���ᷢ�����кͷ�Ӧ���Ӳ�ͬ�Ƕ����������ʵ�鷽����������ʵ�飮
����һ������pH��ֽ�ⶨNaOH��Һ��pH���ٵμ����ᣬ����������Һ��ͬʱ�ⶨ�����Һ��pH�������õ�pH��С��С��7����֤��NaOH��Һ��ϡ���ᷢ���˻�ѧ��Ӧ��
��1����pH��ֽ�ⶨ��Һ��pHʱ����ȷ�IJ����ǣ� ��
��2������ǿ������õ�pHС��7�������ɣ� ��
������������NaOH��Һ�еμӼ��η�̪��Һ����Һ�Ժ�ɫ��Ȼ���ٵμ����ᣬ�ɹ۲쵽��ɫ����ʧ����֤��NaOH��Һ��ϡ���ᷢ���˻�ѧ��Ӧ��
����ͬѧ����NaOH��Һ�еμӷ�̪��Һʱ��������һ��������������������Һ�е����̪��Һ����Һ����˺�ɫ������һ�����ɫ����ʧ�ˣ���С����������������ԭ���������²��룺�ٿ����Ƿ�̪��Һ������е�������Ӧ��ʹ��ɫ��ʧ���ڿ���������������Һ������еĶ�����̼��Ӧ��ʹ��ɫ��ʧ��
��1��Ϊ��֤����٣�����ͬѧ��������ʵ�飺�����Ƶ�����������Һ���ȣ�����Һ���Ϸ���һЩֲ���ͣ�Ȼ������ȴ�����Һ�е����̪��Һ��ʵ���С����ȡ��͡�����ֲ���͡�Ŀ���� ��ʵ����������̪��Һ��ɫ��ʧ������е������أ�
��2��Ϊ��֤����ڣ�����ͬѧ��������ʵ�飺ȡ��һ������Na2CO3��Һ�������е����̪��Һ��������ҺҲ���ֺ�ɫ���ɴ˿ɵó�����������ۣ�����1��˵��Na2CO3��Һ�� �ԣ�����2��˵����̪��Һ��ɫ��ʧ������еĶ�����̼�أ�
��3����С��ͬѧͨ���������ϵ�֪��������������ҺŨ�Ⱥܴ�ʱ���ͻ���������������������ʵ��֤���÷�����ȡ�õ�NaOH��ҺŨ�ȹ���ʵ�鷽�� ���ڹ۲쵽������ ��
����������ѧ��Ӧ��ͨ�������������ı仯���ɽ�����Ӧǰ����¶ȱ仯���жϷ�Ӧ�ķ��������NaOH��Һ��ϡ������ǰ���¶��б仯����֤�������˻�ѧ��Ӧ��
����ͬѧ����ͬŨ�ȵ������NaOH��Һ��10mL��ϣ�����
�ȼƲⶨ�����»��ǰ���¶ȵı仯������¼��ÿ�λ��ǰ���¶ȵ�����ֵ��t���������
��1������x= ��
��2��ijͬѧ��ûʹ���¶ȼƵ�����£�ͨ����ͼ��ʾװ�������ʵ�飮���ͬѧ���� �ж�NaOH��Һ��ϡ���ᷢ�����кͷ�Ӧ��
��3������ʵ���е�ϸ�ں������������ʵ���У�ϡ��������ý�ͷ�ι���εμӣ���������Ŀ���� ����ʵ������У�Ҫ�ò��������Ͻ��裬��������Ŀ���� ������ʵ����������ⷢ�������ݳ��֣�����Ϊԭ���� ���ܷ���ʢ��NaOH��Һ���Լ�ƿƿ�ں���Ƥ���ϳ����˰�ɫ��ĩ������������ѧ�Ļ�ѧ֪ʶ�������ְ�ɫ��ĩ�ijɷ��������²��룺�ٿ�����NaOH������Ϊ���ڿ����� �� �ۿ����� ��
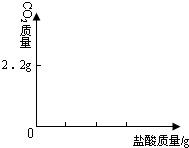
��4��Ϊ�˽�һ���о�ʵ���г��ֵ����⣬ȡ��13.3g�������ƹ�����Ʒ��������ˮ�����Һ�������м���200g10%��ϡ���ᣬʹ���ַ�Ӧ�����ɶ�����̼2.2g����
��1����Ʒ���������Ƶ�������
��2�����������Ʒ�Ӧ�������������
��3����ͼ�л������������ʾ������̼�������������ʾ����������Ĺ�ϵͼ��
����֪Na2CO3+2HCl�T2NaCl+H2O+CO2����
����һ������pH��ֽ�ⶨNaOH��Һ��pH���ٵμ����ᣬ����������Һ��ͬʱ�ⶨ�����Һ��pH�������õ�pH��С��С��7����֤��NaOH��Һ��ϡ���ᷢ���˻�ѧ��Ӧ��
��1����pH��ֽ�ⶨ��Һ��pHʱ����ȷ�IJ����ǣ�
��2������ǿ������õ�pHС��7�������ɣ�
������������NaOH��Һ�еμӼ��η�̪��Һ����Һ�Ժ�ɫ��Ȼ���ٵμ����ᣬ�ɹ۲쵽��ɫ����ʧ����֤��NaOH��Һ��ϡ���ᷢ���˻�ѧ��Ӧ��
����ͬѧ����NaOH��Һ�еμӷ�̪��Һʱ��������һ��������������������Һ�е����̪��Һ����Һ����˺�ɫ������һ�����ɫ����ʧ�ˣ���С����������������ԭ���������²��룺�ٿ����Ƿ�̪��Һ������е�������Ӧ��ʹ��ɫ��ʧ���ڿ���������������Һ������еĶ�����̼��Ӧ��ʹ��ɫ��ʧ��
��1��Ϊ��֤����٣�����ͬѧ��������ʵ�飺�����Ƶ�����������Һ���ȣ�����Һ���Ϸ���һЩֲ���ͣ�Ȼ������ȴ�����Һ�е����̪��Һ��ʵ���С����ȡ��͡�����ֲ���͡�Ŀ����
��2��Ϊ��֤����ڣ�����ͬѧ��������ʵ�飺ȡ��һ������Na2CO3��Һ�������е����̪��Һ��������ҺҲ���ֺ�ɫ���ɴ˿ɵó�����������ۣ�����1��˵��Na2CO3��Һ��
��3����С��ͬѧͨ���������ϵ�֪��������������ҺŨ�Ⱥܴ�ʱ���ͻ���������������������ʵ��֤���÷�����ȡ�õ�NaOH��ҺŨ�ȹ���ʵ�鷽��
����������ѧ��Ӧ��ͨ�������������ı仯���ɽ�����Ӧǰ����¶ȱ仯���жϷ�Ӧ�ķ��������NaOH��Һ��ϡ������ǰ���¶��б仯����֤�������˻�ѧ��Ӧ��
����ͬѧ����ͬŨ�ȵ������NaOH��Һ��10mL��ϣ�����
�ȼƲⶨ�����»��ǰ���¶ȵı仯������¼��ÿ�λ��ǰ���¶ȵ�����ֵ��t���������
| ��� | ���� | NaOH��Һ | ��t/�� |
| 1 | 3.65% | 2.00% | 3.5 |
| 2 | 3.65% | 4.00% | x |
| 3 | 7.30% | 8.00% | 14 |

��1������x=
��2��ijͬѧ��ûʹ���¶ȼƵ�����£�ͨ����ͼ��ʾװ�������ʵ�飮���ͬѧ����
��3������ʵ���е�ϸ�ں������������ʵ���У�ϡ��������ý�ͷ�ι���εμӣ���������Ŀ����
��4��Ϊ�˽�һ���о�ʵ���г��ֵ����⣬ȡ��13.3g�������ƹ�����Ʒ��������ˮ�����Һ�������м���200g10%��ϡ���ᣬʹ���ַ�Ӧ�����ɶ�����̼2.2g����
��1����Ʒ���������Ƶ�������
��2�����������Ʒ�Ӧ�������������
��3����ͼ�л������������ʾ������̼�������������ʾ����������Ĺ�ϵͼ��

����֪Na2CO3+2HCl�T2NaCl+H2O+CO2����
ij��ȤС��ͬѧΪ֤��NaOH��Һ��ϡ���ᷢ�����кͷ�Ӧ���Ӳ�ͬ�Ƕ����������ʵ�鷽����������ʵ�飮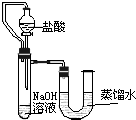
I��ʵ�鷽��
����һ������pH��ֽ�ⶨNaOH��Һ��pH���ٵμ����ᣬ����������Һ��ͬʱ�ⶨ�����Һ��pH�������õ�pH��С��С��7����֤��NaOH��Һ��ϡ���ᷢ���˻�ѧ��Ӧ��
��1����pH��ֽ�ⶨNaOH��ҺpHʱ����ȷ�IJ����ǣ� ��
��2������ǿ������õ�pHС��7�������ɣ� ��
����������ѧ��Ӧ��ͨ�������������ı仯���ɽ�����Ӧǰ����¶ȱ仯���жϷ�Ӧ�ķ��������
NaOH��Һ��ϡ������ǰ���¶��б仯����֤�������˻�ѧ��Ӧ��
����ͬѧ����ͬŨ�ȵ������NaOH��Һ��10�˻�ϣ����¶ȼƲⶨ�����»��ǰ���¶ȵı仯������¼��ÿ�λ��ǰ���¶ȵ�����ֵ��t�����±�����
��1������x= ��
��2��ijͬѧ��ûʹ���¶ȼƵ�����£�ͨ����ͼ��ʾװ�������ʵ�飮���ͬѧ���� �ж�NaOH��Һ��ϡ���ᷢ�����кͷ�Ӧ��
II��ʵ���е��������
��ʵ������У����Ƿ���ʢ��NaOH��Һ���Լ�ƿƿ�ں���Ƥ���ϳ����˰�ɫ��ĩ������������ѧ�Ļ�ѧ֪ʶ�������ְ�ɫ��ĩ�ijɷ��������²��룺�ٿ�����NaOH���ڿ�����Na2CO3�� �ۿ�����NaOH��Na2CO3��
��1��С��ͬѧȡ��ɫ��ĩ����������ˮ��������Һ�м��������� ��Һ��������ɫ����������Ȼ�����ϲ���Һ�м����̪��Һ��������Һ�ʺ�ɫ����֤�˲��������ȷ�ģ�
��2��Ϊ�˽�һ���о�����λͬѧȡ��10.0g������Ʒ�����õ�����ƽ��ͬ������ͼ��ʾ��ʵ�飮
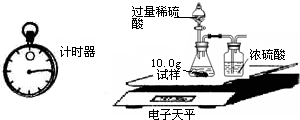
ʵ�����ݼ�¼���£�
��ͨ�������������ݼ���������Ʒ�и��ɷݵ�����������д��������̣��� ��
��3����ͬѧ���������ʵ�������������Na2CO3����������ƫС����ͬѧ�������ǣ�ʵ���������ȷ���� ��

I��ʵ�鷽��
����һ������pH��ֽ�ⶨNaOH��Һ��pH���ٵμ����ᣬ����������Һ��ͬʱ�ⶨ�����Һ��pH�������õ�pH��С��С��7����֤��NaOH��Һ��ϡ���ᷢ���˻�ѧ��Ӧ��
��1����pH��ֽ�ⶨNaOH��ҺpHʱ����ȷ�IJ����ǣ�
��2������ǿ������õ�pHС��7�������ɣ�
����������ѧ��Ӧ��ͨ�������������ı仯���ɽ�����Ӧǰ����¶ȱ仯���жϷ�Ӧ�ķ��������
NaOH��Һ��ϡ������ǰ���¶��б仯����֤�������˻�ѧ��Ӧ��
����ͬѧ����ͬŨ�ȵ������NaOH��Һ��10�˻�ϣ����¶ȼƲⶨ�����»��ǰ���¶ȵı仯������¼��ÿ�λ��ǰ���¶ȵ�����ֵ��t�����±�����
| ��� | ���� | NaOH��Һ | ��t/�� |
| 1 | 3.65% | 2.00% | 3.5 |
| 2 | 3.65% | 4.00% | x |
| 3 | 7.30% | 8.00% | 14 |
��2��ijͬѧ��ûʹ���¶ȼƵ�����£�ͨ����ͼ��ʾװ�������ʵ�飮���ͬѧ����
II��ʵ���е��������
��ʵ������У����Ƿ���ʢ��NaOH��Һ���Լ�ƿƿ�ں���Ƥ���ϳ����˰�ɫ��ĩ������������ѧ�Ļ�ѧ֪ʶ�������ְ�ɫ��ĩ�ijɷ��������²��룺�ٿ�����NaOH���ڿ�����Na2CO3�� �ۿ�����NaOH��Na2CO3��
��1��С��ͬѧȡ��ɫ��ĩ����������ˮ��������Һ�м���������
��2��Ϊ�˽�һ���о�����λͬѧȡ��10.0g������Ʒ�����õ�����ƽ��ͬ������ͼ��ʾ��ʵ�飮

ʵ�����ݼ�¼���£�
| �� �� �� Ŀ | �� �� ʱ �� | ������g�� |
| ���� | 10.00 | |
| װ��+ϡ�������� | 241.20 | |
| װ��+ϡ��������+���� | ��Ӧ��ʼ��15�� | 249.20 |
| װ��+ϡ��������+���� | ��Ӧ��ʼ��35�� | 249.00 |
| װ��+ϡ��������+���� | ��Ӧ��ʼ��55�� | 249.00 |
��3����ͬѧ���������ʵ�������������Na2CO3����������ƫС����ͬѧ�������ǣ�ʵ���������ȷ����