��Ŀ����
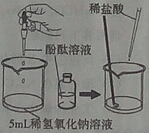
��8�֣�ijͬѧ����ʵ��ʱ��֪�Ȼ�����Ʒ�к���������ɳ���Ȼ��ƺ��Ȼ�þ������������������̳�ȥ�Ȼ�����Ʒ����ɳ���Ȼ��ƺ��Ȼ�þ��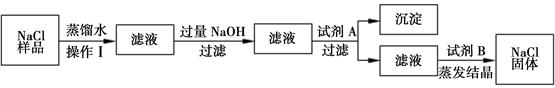
���������ͼ�ش�
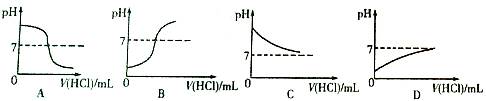
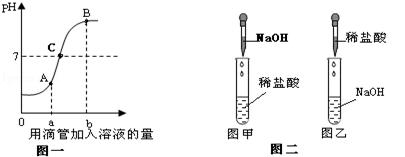
��1��������������� ���Լ�A�� ���Լ�B�� ��
��2��д��NaOH�����ʷ�Ӧ�Ļ�ѧ����ʽ ��
��3�������ᾧʱ�������������� ��
��4�����ᴿ����Ȼ��ƹ���������50g��������Ϊ6%���Ȼ�����Һ���䲽��Ϊ���ټ��㡢�ڳ������� ���� �ܽ⡢��װ���Լ�ƿ�У��Ǻ�ƿ���� ��
��1������;Na2CO3 ; HCl��2��2NaOH+MgCl2=Mg(OH)2��+2NaCl��3�����裬��ֹ��ֲ��¶ȹ��ߣ����Һ�ηɽ���4����ȡ ���ϱ�ǩ
����

��5�֣������±����ݣ��ش��������⡣
| ���� | �������� | �״� | ʳ��ˮ | ϴ�Ӽ� | ¯������ |
| pH | 1 | 3 | 7 | 9 | 12 |
��2��ˮ���е�ˮ������Ҫ�ɷ�Ϊ̼��ƺ�������þ���ð״� ����ܡ����ܡ���ȥ����
��3����������ϴ�Ӽ���ϴ�;��ϵ����ۣ������������� ��
��4������������¯������ ����ܡ����ܡ������ʹ�ã��ɱ���֪ʳ��ˮ�����ԣ��پٳ�һ�ֳ����Ե���Һ���� ��Һ��
���ҡ���һ����ѧС���磬�̺��ḻ�Ļ�ѧ֪ʶ��
�� ̼�������г�������������������ᣨC6H5COOH�����÷������� ��1�� ��Ԫ����ɣ����� ��2�� ����л���������������̼����Ԫ�ص�ԭ�Ӹ�����Ϊ ��3�� ��
�� ��ʯ������ʳƷ��������仯ѧʽΪ ��4�� ���ȵĴ�����Һ��ϴ�����ۣ�����Ļ�ѧʽΪ ��5�� ��
�� �±�Ϊ���ּ��������Ĺ��ܼ���Ч�ɷ֣�
| �� �� | ����� | ������Ư | Ư�� |
| �� �� | ��Ч����۹�����ζ | Ưϴʹ����ɫ������ | ����Ư���������� |
| ��Ч�ɷ� | HCl | H2O2 | NaClO |
�����ⶨ������顱�����ȣ���ѡ�� ��7�� ������ĸѡ���
A��pH��ֽ B����̪��Һ C��ʯ����Һ
��������Ư��Һ���м��������������̺�����������ݣ�д���÷�Ӧ�Ļ�ѧ����ʽ ��8�� ��
����������顱�롰Ư�������ܻ��ã�������ײ���һ���ж�����X����Ӧ
ԭ��Ϊ��NaClO + 2HCl
 NaCl + X�� + H2O ����X�Ļ�ѧʽΪ ��9�� ��
NaCl + X�� + H2O ����X�Ļ�ѧʽΪ ��9�� ��