��Ŀ����
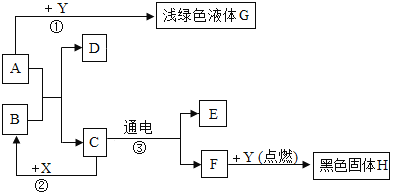
����Ŀ����Ȼ���ж�����̼��ѭ��������ͼ��ʾ��
��1�����մ�����CO2�Ļ�����________________________��
��2�������������Ļ�ʯȼ�ϵ����Ӻ�ɭ�ֵ��ҿ��ķ���ʹ������CO2�ĺ����������Ӷ�����___________________��ǿ��ȫ�������ů����д��һ���������ů���������滷������Ӱ��������___________________________________________________��
��3����ѧ���о��õ������������մ���CO2�ĵط��������ҵ����������CO2�ŷ���������Ŀǰ�ٶ����ӣ�����Ϊ���ữ�ĺ���������д���ữԭ��Ļ�ѧ��Ӧ����ʽ��___________________��
��4����ѧ��Ŀǰ�����о��������й�����CO2��H2�ڴ���������������ת����Һ̬�״���ˮ����Ӧ�Ļ�ѧ����ʽ��CO2+3H2![]() �״�+H2O����״��Ļ�ѧʽ��___��
�״�+H2O����״��Ļ�ѧʽ��___��
���𰸡���ɫֲ��Ĺ������ �������յ�̫��������� ���������ı����ڻ���ʹ��ƽ�����ߣ���û�����غ����У��Լ�ʹ����ɳĮ����ũҵ������ CO2+H2O=H2CO3 CH3OH
��������
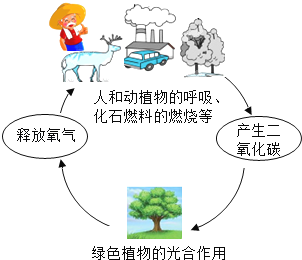
��1����ɫֲ��Ĺ�����ÿ������ն�����̼���ͷ�������
��2��������̼�����������ҵIJ��������ϱ�Ĥ������ʹ�������յ�̫�������������ɢʧ���Ӷ�ʹȫ���ů�����������������ЧӦ����ȫ���ů���ܵ��������ı����ڻ���ʹ��ƽ�����ߣ���û�����غ����У��Լ�ʹ����ɳĮ����ũҵ�����ȣ�
��3��������̼��ˮ��Ӧ����̼�ᣬ��ѧ��Ӧ����ʽ��CO2+H2O=H2CO3��
��4�����ݻ�ѧ��Ӧǰ��ԭ�ӵ��������Ŀ���䣬��CO2+3H2![]() �״�+H2O��֪���״��Ļ�ѧʽ��CH3OH��
�״�+H2O��֪���״��Ļ�ѧʽ��CH3OH��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�����Ŀ������ͭ��Һ�Թ�������ķֽ��д����ã�ȡһ������8.5%�Ĺ���������Һ�����ձ��У�����һ������15%������ͭ��Һ������������ȫ�ֽ⣬�й�ʵ�����������ʾ��������������ˮ�е��ܽ⣩������㣺
��Ӧǰ | ��Ӧ�� | ||
ʵ������ | �ձ�����������Һ������/g | ����ͭ��Һ������/g | �ձ����ձ�����Һ������/g |
60 | 3.3 | 62.5 | |
��1����������������Ϊ________��
��2����Ӧ���ձ�����Һ���ʵ�������������д��������̣������ȷ��0.1%����