��Ŀ����
����Ŀ�������д����л�ѧ��������ѧ֪ʶ�ش��������⣺
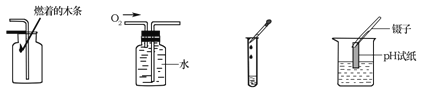
��1��ijѧУʳ�õ�����ṩ��������С���ࡢ���������ͷ������������Ӿ���Ӫ���ĽǶȽ���ʳ�����Ӻ�___________________��ʳ�����ĸ����
A�������� B����֬ C������ D��ά����
��2���״���CH3OH���ж���������ʹ�۾�ʧ�������������������о�֤�����ð�����NH3���������м״��Ĺ�ҵ��ˮ����ʹ��ת����������ʣ��йط�Ӧ�Ļ�ѧ����ʽΪ��5CH3OH+12O2+6NH3![]() 3X+5CO2+19H2O����X�Ļ�ѧʽΪ________________��
3X+5CO2+19H2O����X�Ļ�ѧʽΪ________________��
��3�����飨C3H8���Ǽ���Һ��ʯ��������Ҫ�ɷ�֮һ�����ͼ����ڿ�����ȼ�յ���������ͬ����д��������ȼ�յĻ�ѧ����ʽ_______________________��
���𰸡� D N2 C3H8+5O2![]() 3CO2+4H2O
3CO2+4H2O
����������1��Уʳ�õ�����ṩ���������������ʡ�С���������Ӫ���ء���������������ʺ�֬������ͷ�������������ۣ��Ӿ���Ӫ���ĽǶȽ���ʳ�����Ӻ�ά���ص�ʳ����ۡ������ʣ���֬��ά���ض�����������Ӫ��Ԫ�ء���2���״���CH3OH���ж���������ʹ�۾�ʧ���������������ð�����NH3���������м״��Ĺ�ҵ��ˮ����ʹ��ת����������ʣ��йط�Ӧ�Ļ�ѧ����ʽΪ��5CH3OH+12O2+6NH3![]() 3X+5CO2+19H2O�����������غ㶨��֪����X�Ļ�ѧʽΪN2���κ�һ����ѧ��Ӧ����Ȼ��ѭ�����غ㶨�ɡ���3�����飨C3H8��
3X+5CO2+19H2O�����������غ㶨��֪����X�Ļ�ѧʽΪN2���κ�һ����ѧ��Ӧ����Ȼ��ѭ�����غ㶨�ɡ���3�����飨C3H8��
�ͼ����ڿ�����ȼ�յ���������ͬ��������ȼ�յĻ�ѧ����ʽ�� C3H8+5O2![]() 3CO2+4H2O��
3CO2+4H2O��
�㾦�ü״���CH3OH���ж���������ʹ�۾�ʧ���������������κ�һ����ѧ��Ӧ����Ȼ��ѭ�����غ㶨�ɡ�

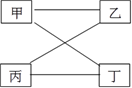
 �Ķ��쳵ϵ�д�
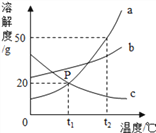
�Ķ��쳵ϵ�д�����Ŀ���±��ṩ���������ʷ���ͼʾ�������ʼ���һ�������¿��Է�����Ӧ����
�� | �� | �� | �� | |
A | CO | O2 | H2SO4 | CuO |
B | Mg | HCl | CuSO4 | NaOH |
C | KOH | SO3 | NaOH | CuSO4 |
D | Na2CO3 | Ca(OH)2 | HCl | CaCl2 |
A. A B. B C. C D. D
����Ŀ�����ݵ���ʯ��ʯ��Դ�ḻ��ij��ѧ��ȤС���ȡ4.0gʯ��ʯ��Ʒ����40gϡ�����4�μ�����Ʒ��(�������ʲ���ӦҲ���ܽ�)����ʵ���������£�
ϡ��������� | ʣ���������� |
��һ�μ���10g | 3.0g |
�ڶ��μ���10g | 2.0g |
�������10g | 1.0g |
���Ĵμ���10g | 0.6g |
����㣺
(1)4.0gʯ��ʯ��Ʒ��̼��Ƶ�������____g��
(2)10gϡ��������_____g̼���������ȫ��Ӧ��
(3)��ϡ�����������������Ϊ_________(д��������̣������ȷ��0.1%)