��Ŀ����
��ϰ���ϣ���ʦ������ƿû�б�ǩ�İ�ɫ���壬�ֱ���̼���ơ��������ơ��Ȼ�狀��Ȼ��ƣ�Ҫ��ͬѧ�����ʵ�鷽�����м���С���������ͼ��ʾ�ķ���������ʵ�飨ʵ�������ÿһ�����ȡ��ԭ�������ʣ���
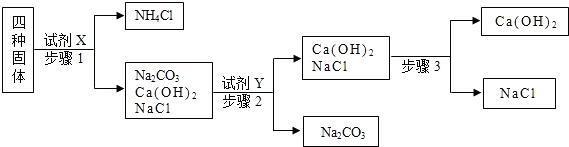
��1���������Լ�X����ʯ�ң���ʯ�����Ȼ�炙�ϵ�ʵ�������� ��д����ʯ����NH4Cl��Ӧ�Ļ�ѧ����ʽ ��
��2�������Լ�Y������ʵ������ȡCO2����Y�Ļ�ѧʽ�� ���Լ�Y�뱻����������ʷ�Ӧ�Ļ�ѧ����ʽΪ�� ��
��3������3�зֱ�ȡ���������ֹ�������ˮ������Һ��pH���������������ʣ���pH��ֽ����ҺpH�IJ����ǣ�ȡһСƬpH��ֽ���ڲ���Ƭ�ϣ� ������pH��ֵ��

��1���������Լ�X����ʯ�ң���ʯ�����Ȼ�炙�ϵ�ʵ��������
��2�������Լ�Y������ʵ������ȡCO2����Y�Ļ�ѧʽ��
��3������3�зֱ�ȡ���������ֹ�������ˮ������Һ��pH���������������ʣ���pH��ֽ����ҺpH�IJ����ǣ�ȡһСƬpH��ֽ���ڲ���Ƭ�ϣ�
���㣺���ʵļ����ƶ�,��Һ�����Ȳⶨ,��Ļ�ѧ����,�εĻ�ѧ����,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺���ʵļ�����
��������1�������Ȼ�狀��������Ʒ�Ӧ�������д̼�����ζ�İ������з�����
��2������ʵ������ȡ������̼��ҩƷ�������ʯ��ʯ���з�����
��3������ʵ������pH��ֽ�ⶨ��ҺpHֵ����ȷ�����������з�����
��2������ʵ������ȡ������̼��ҩƷ�������ʯ��ʯ���з�����
��3������ʵ������pH��ֽ�ⶨ��ҺpHֵ����ȷ�����������з�����
����⣺��1���Ȼ�狀��������Ʒ�Ӧ�������д̼�����ζ�İ�����ˮ���Ȼ��ƣ�����ʵ�������ǣ��д̼�����ζ�������������ѧ����ʽΪ��Ca��OH��2+2NH4Cl=CaCl2+2H2O+2NH3����
��2��ʵ������ȡ������̼��ҩƷ�������ʯ��ʯ��Y����̼���Ʒ�Ӧ������Y�����ᣬ��ѧʽ�ǣ�HCl�������̼���Ʒ�Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼����ѧ����ʽΪ��Na2CO3+2HCl=2NaCl+H2O+CO2����
��3����pH��ֽ����ҺpH�IJ����ǣ�ȡһСƬpH��ֽ���ڲ���Ƭ�ϣ��ò�����պȡ��������Һ��ε�pH��ֽ�ϣ�Ȼ����ʾ��ɫ�����ɫ�����գ�����pH��ֵ��
�ʴ�Ϊ����1���д̼�����ζ�����������Ca��OH��2+2NH4Cl=CaCl2+2H2O+2NH3����
��2��HCl��Na2CO3+2HCl=2NaCl+H2O+CO2����
��3���ò�����պȡ��������Һ��ε�pH��ֽ�ϣ�Ȼ����ʾ��ɫ�����ɫ�����գ�
��2��ʵ������ȡ������̼��ҩƷ�������ʯ��ʯ��Y����̼���Ʒ�Ӧ������Y�����ᣬ��ѧʽ�ǣ�HCl�������̼���Ʒ�Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼����ѧ����ʽΪ��Na2CO3+2HCl=2NaCl+H2O+CO2����
��3����pH��ֽ����ҺpH�IJ����ǣ�ȡһСƬpH��ֽ���ڲ���Ƭ�ϣ��ò�����պȡ��������Һ��ε�pH��ֽ�ϣ�Ȼ����ʾ��ɫ�����ɫ�����գ�����pH��ֵ��
�ʴ�Ϊ����1���д̼�����ζ�����������Ca��OH��2+2NH4Cl=CaCl2+2H2O+2NH3����
��2��HCl��Na2CO3+2HCl=2NaCl+H2O+CO2����
��3���ò�����պȡ��������Һ��ε�pH��ֽ�ϣ�Ȼ����ʾ��ɫ�����ɫ�����գ�
���������⿼���˳������ʵļ�����ɴ��⣬�����������ʵ����ʲ�����У�

��ϰ��ϵ�д�
�����Ŀ
�ڻ�ҩ���ҹ��Ĵ���֮һ���ڻ�ҩ�ı�ը����2KNO3+3C+S�TK2S+N2+3X��ʾ����X�Ļ�ѧʽΪ��������
| A��CO |
| B��SO2 |
| C��NO |
| D��CO2 |
 ͨ����ѧѧϰ�������γ���һЩ��ѧ�������
ͨ����ѧѧϰ�������γ���һЩ��ѧ������� ������̼ԭ�ӣ���
������̼ԭ�ӣ��� ��������ԭ�ӣ���
��������ԭ�ӣ���
 �������ӵĺ˵����Ϊ
�������ӵĺ˵����Ϊ