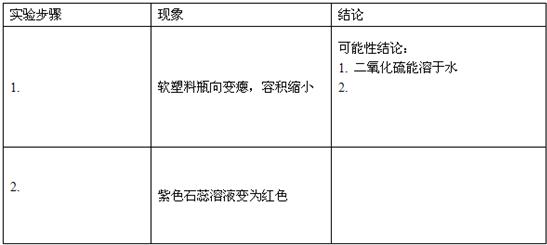
��Ŀ����
��ʵ�������ʦ����ͼ��ʾ��װ�ý���ʵ�飨ͼ�й̶��Թܵ���������ȥ��������һ��ʱ��۲쵽ľ̿�۵��������Լ��٣�����ͭ���²�������һ�������ɫ�����ʣ�����ʯ��ˮ����ǣ�ֹͣ���Ȳ���ȴ�����£�
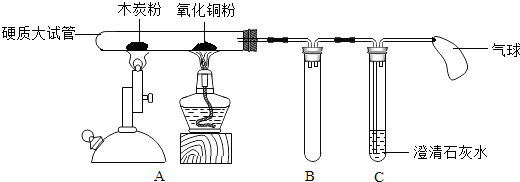
��1��Ӳ�ʴ��Թ�������ͭ������Ӧ�Ļ�ѧ����ʽ��
��2��Bװ�õ�������
��3��Cװ���з�����Ӧ�Ļ�ѧ����ʽ��

��1��Ӳ�ʴ��Թ�������ͭ������Ӧ�Ļ�ѧ����ʽ��
CuO+CO
Cu+CO2
| ||
CuO+CO
Cu+CO2
��
| ||
��2��Bװ�õ�������
��ֹֹͣ���Ȳ���ȴ������ʱ��װ��C�е�Һ�嵹����װ��A��Ӳ�ʴ��Թ��У�ը���Թ�
��ֹֹͣ���Ȳ���ȴ������ʱ��װ��C�е�Һ�嵹����װ��A��Ӳ�ʴ��Թ��У�ը���Թ�
����3��Cװ���з�����Ӧ�Ļ�ѧ����ʽ��
CO2+Ca��OH��2�TCaCO3��+H2O
CO2+Ca��OH��2�TCaCO3��+H2O
����������1������������Ӳ���Թ������ʵ����ʷ�����Ӧ�ķ�����
��2����Ӧ��������������ȴ��������ѹǿ��С��Cװ���е���Һ�ᵹ��������Bװ�ÿɷ�ֹ��Һ����Ӳ�ʴ��Թ��У�
��3�����ݶ�����̼���������Ʒ�Ӧ����������ȷд����ѧ����ʽ��
��2����Ӧ��������������ȴ��������ѹǿ��С��Cװ���е���Һ�ᵹ��������Bװ�ÿɷ�ֹ��Һ����Ӳ�ʴ��Թ��У�
��3�����ݶ�����̼���������Ʒ�Ӧ����������ȷд����ѧ����ʽ��
����⣺��1����Ӳ���Թ��к���̼������ͭ��������̼����������һ����̼��һ����̼�ɻ�ԭ����ͭ��������ͭ�μӷ�Ӧ�Ļ�ѧ����ʽΪ��CuO+CO
Cu+CO2��
��2����Ӧ��������������ȴ��������ѹǿ��С��Cװ���е���Һ�ᵹ��������Bװ�ÿɷ�ֹ��Һ������Ӳ�ʴ��Թ��У�
��3����Ӧ�����˶�����̼��������̼��ʹ����ʯ��ˮ����ǣ���ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O��
�ʴ�Ϊ��
��1��CuO+CO
Cu+CO2��
��2����ֹֹͣ���Ȳ���ȴ������ʱ��װ��C�е�Һ�嵹����װ��A��Ӳ�ʴ��Թ��У�ը���Թܣ�
��3��CO2+Ca��OH��2�TCaCO3��+H2O��
| ||
��2����Ӧ��������������ȴ��������ѹǿ��С��Cװ���е���Һ�ᵹ��������Bװ�ÿɷ�ֹ��Һ������Ӳ�ʴ��Թ��У�
��3����Ӧ�����˶�����̼��������̼��ʹ����ʯ��ˮ����ǣ���ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O��
�ʴ�Ϊ��
��1��CuO+CO
| ||
��2����ֹֹͣ���Ȳ���ȴ������ʱ��װ��C�е�Һ�嵹����װ��A��Ӳ�ʴ��Թ��У�ը���Թܣ�
��3��CO2+Ca��OH��2�TCaCO3��+H2O��
������������һ��ʵ����ۺ��⣬����Ĺؼ��Ƕ�����������ʵķ������������ʱʵ���һЩ�ؼ�����Ҳ�����ǽ������Ҫͻ�ƿڣ�

��ϰ��ϵ�д�
 ��������ϵ�д�
��������ϵ�д�
�����Ŀ
 27����ѧʵ�������ʦ��ͬѧ����������ʵ��װ�ã�
27����ѧʵ�������ʦ��ͬѧ����������ʵ��װ�ã�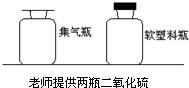 23�������Ǻ�С��һ���߽���ѧʵ���ң���ͬ��ѧϰ��ѧ̽���ķ�����
23�������Ǻ�С��һ���߽���ѧʵ���ң���ͬ��ѧϰ��ѧ̽���ķ�����