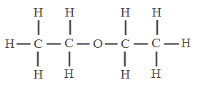��Ŀ����
����Ŀ����ѧ��ȤС��������ʦ��ָ���£������ϸ���֭Һ�����кͷ�Ӧʵ�顣
����ʾʵ�顿ȡ�������õ�NaOH��Һ�������뼸���ϸ���֭Һ����Һ���ϱ�ƣ���Ȼ�����ɫNaOH��Һ����μ���ϡ���ᣬ������Һ��ð���ݣ�����Һ�����������γ��ֻ�ɫ����ɫ����ɫ����ɫ���ۺ졢���ɫ�ȣ������ʺ硢�dz�������
̽����Ϊʲô��Ӧ��������Һ����ֶ�ʵ���ɫ
�����в��롿
Сͮ���룺�����Ƿ�Ӧ�����У���Һ�и���λ�ϸ���֭Һ��Ũ�Ȳ�ͬ��
С�ܲ��룺�����Ƿ�Ӧ�����У���Һ�и���λ�����ȣ�pH����ͬ��
��ʵ����֤��
��1����С�ձ��м��������ϸ���֭Һ��Ȼ������������ˮ����Һʼ������ɫ��
��2������һ��С�ձ��м������������Ƶ�NaOH��Һ��Ȼ����μ���ϡ���ᣬ��ʱ�۲���Һ��ɫ�ı仯��ͬʱ�ô����������Ӧ��������ҺpH�ı仯��������ҺpH����ɫ�ı仯ͼ����ͼ��ʾ������ͼ��֪��______________________�IJ�������ȷ�ġ�

����չ���졿���ͼ��˼����
��1�������ǡ����ȫ�к�ʱ����Һ��____________________ɫ��
��2������Һ�ʺ�ɫʱ����Ӧ��Һ�е�������________________��
̽�����õ�NaOH��Һ�����ʳɷ�
���������ϡ�CaCl2��Һ�����ԡ�
��ʵ����֤��
��1��С����Ϊ����Ϊ����õ�NaOH��Һ�еμ�ϡ����ʱ���������ݣ����Ը���Һ�ѱ��ʡ����û�ѧ����ʽ��ʾNaOH��Һ���ʵ�ԭ��___________________________________��
��2��С��ȡ�������õ�NaOH��Һ����������CaCl2��Һ��������ɫ�����������ϲ���Һ�е��뼸���ϸ���֭Һ����Һ�Ի�ɫ����ˣ���ƿ���õ�NaOH��Һ�����ʳɷ���_____________��
�����۷�˼����Ϊ���ָʾ�����ϸ���֭Һ����ɫʯ����Һ��ȣ��ŵ����ԣ������ܲ����Һ������ԣ�������ʾ��Һ��___________________________________________��
���𰸡� С�� �� NaCl��HCl CO2+2NaOH�TNa2CO3+H2O NaOH��Na2CO3 �����ǿ�������Ȼ�һ����Χ�ڵ�pH
��������������ѧ֪ʶ��������Ϣ֪������ʾʵ�顿ȡ�������õ�NaOH��Һ�������뼸���ϸ���֭Һ����Һ���ϱ�ƣ���Ȼ�����ɫNaOH��Һ����μ���ϡ���ᣬ������Һ��ð���ݣ�����Һ�����������γ��ֻ�ɫ����ɫ����ɫ����ɫ���ۺ졢���ɫ�ȣ������ʺ硢�dz�������̽����Ϊʲô��Ӧ��������Һ����ֶ�ʵ���ɫ�������в��롿Сͮ���룺�����Ƿ�Ӧ�����У���Һ�и���λ�ϸ���֭Һ��Ũ�Ȳ�ͬ��С�ܲ��룺�����Ƿ�Ӧ�����У���Һ�и���λ�����ȣ�pH����ͬ����ʵ����֤����1����С�ձ��м��������ϸ���֭Һ��Ȼ������������ˮ����Һʼ������ɫ����2������һ��С�ձ��м������������Ƶ�NaOH��Һ��Ȼ����μ���ϡ���ᣬ��ʱ�۲���Һ��ɫ�ı仯��ͬʱ�ô����������Ӧ��������ҺpH�ı仯��������ҺpH����ɫ�ı仯ͼ����ͼ��ʾ������ͼ��֪��С�ܵIJ�������ȷ�ġ�����չ���졿���ͼ��˼������1�������ǡ����ȫ�к�ʱ����Һ����ɫ����2������Һ�ʺ�ɫʱ����Ӧ��Һ�е�������NaCl��HCl���������̽�����õ�NaOH��Һ�����ʳɷ֡����������ϡ�CaCl2��Һ�����ԡ���ʵ����֤����1��С����Ϊ����Ϊ����õ�NaOH��Һ�еμ�ϡ����ʱ���������ݣ����Ը���Һ�ѱ��ʡ����û�ѧ����ʽ��ʾNaOH��Һ���ʵ�ԭ��CO2+2NaOH�TNa2CO3+H2O ����2��С��ȡ�������õ�NaOH��Һ����������CaCl2��Һ��������ɫ�����������ϲ���Һ�е��뼸���ϸ���֭Һ����Һ�Ի�ɫ����ˣ���ƿ���õ�NaOH��Һ�����ʳɷ���NaOH��Na2CO3 �����ֱ��� �������۷�˼����Ϊ���ָʾ�����ϸ���֭Һ����ɫʯ����Һ��ȣ��ŵ����ԣ������ܲ����Һ������ԣ�������ʾ��Һ�� �����ǿ�������Ȼ�һ����Χ�ڵ�pH��
�㾦��������Ҫ�����кͷ�Ӧʵ�顣

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�