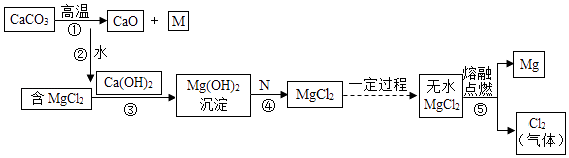
��Ŀ����
����Ŀ��̼Ԫ��������������ʵĻ���Ԫ�أ��ش����к�̼Ԫ�����ʵ��й����⣮
��1����������̼�������Ҫ��Ϊ�˼������ŷ�����
��2������CO2���ó���ʯ��ˮ��д���÷�Ӧ�Ļ�ѧ����ʽ ��
��3����Ȼ������Ҫ�ɷ���CH4����ȼ�����û�ѧ����ʽ��ʾ����
��4��Ϊ������������CO2���ŷţ���ѧ�ҽ�CO2��H2�ڴ����ͼ��ȵ������·�Ӧ��ת��Ϊˮ�ͼ��飬�÷�Ӧ�Ļ�ѧ����ʽΪ �� ����˵����������CO2�ĺ���Խ��Խ�á������Ƿ�ͬ������˵������ǡ��� �� ������ ��
��5�����ʯ��ʯī���������ʲ���ϴ���Ҫԭ���� ��
��6���ڹ�ҵ�Ͽ�����CO��NaOHͨ�����Ϸ�Ӧ�Ʊ�HCOONa�������ƣ����÷�Ӧ�Ļ�ѧ����ʽΪ ��
���𰸡�
��1��CO2
��2��CO2+Ca��OH��2�TCaCO3��+H2O
��3��CH4+2O2 ![]() CO2+2H2O
CO2+2H2O
��4��CO2+4H2 ![]() CH4+2H2O������������ж�����̼�ĺ���̫�ͣ���Ӱ��ֲ��Ĺ������
CH4+2H2O������������ж�����̼�ĺ���̫�ͣ���Ӱ��ֲ��Ĺ������
��5��̼ԭ�ӵ����з�ʽ��ͬ
��6��CO+NaOH ![]() HCOONa
HCOONa
���������⣺��1������̼��������������������ģ���Ҫ��Ϊ�˼��ٶ�����̼���ŷţ���2��������̼���������Ʒ�Ӧ����̼��Ƴ�����ˮ����Ӧ�Ļ�ѧ����ʽΪ��CO2+Ca��OH��2�TCaCO3��+H2O����3�������������ڵ�ȼ�������·�Ӧ���ɶ�����̼��ˮ����ѧ����ʽ��ʾΪ��CH4+2O2 ![]() CO2+2H2O����4��������̼�������ڴ����ͼ��ȵ������·�Ӧ����ˮ�ͼ��飬��ѧ����ʽΪ��CO2+4H2
CO2+2H2O����4��������̼�������ڴ����ͼ��ȵ������·�Ӧ����ˮ�ͼ��飬��ѧ����ʽΪ��CO2+4H2 ![]() CH4+2H2O�� ����˵����������CO2�ĺ���Խ��Խ�á�����ͬ������˵���������ǣ���������ж�����̼�ĺ���̫�ͣ���Ӱ��ֲ��Ĺ�����ã���5�����ʯ��ʯī���������ʲ���ϴ���Ҫԭ����̼ԭ�ӵ����з�ʽ��ͬ����6���ڹ�ҵ�Ͽ�����CO��NaOHͨ�����Ϸ�Ӧ�Ʊ�HCOONa�������ƣ����÷�Ӧ�Ļ�ѧ����ʽΪ��CO+NaOH
CH4+2H2O�� ����˵����������CO2�ĺ���Խ��Խ�á�����ͬ������˵���������ǣ���������ж�����̼�ĺ���̫�ͣ���Ӱ��ֲ��Ĺ�����ã���5�����ʯ��ʯī���������ʲ���ϴ���Ҫԭ����̼ԭ�ӵ����з�ʽ��ͬ����6���ڹ�ҵ�Ͽ�����CO��NaOHͨ�����Ϸ�Ӧ�Ʊ�HCOONa�������ƣ����÷�Ӧ�Ļ�ѧ����ʽΪ��CO+NaOH ![]() HCOONa
HCOONa
���Դ��ǣ���1��CO2 �� ��2��CO2+Ca��OH��2�TCaCO3��+H2O����3��CH4+2O2 ![]() CO2+2H2O����4��CO2+4H2
CO2+2H2O����4��CO2+4H2 ![]() CH4+2H2O������������ж�����̼�ĺ���̫�ͣ���Ӱ��ֲ��Ĺ�����ã���5��̼ԭ�ӵ����з�ʽ��ͬ����6��CO+NaOH
CH4+2H2O������������ж�����̼�ĺ���̫�ͣ���Ӱ��ֲ��Ĺ�����ã���5��̼ԭ�ӵ����з�ʽ��ͬ����6��CO+NaOH ![]() HCOONa
HCOONa
�����㾫�����������⣬������Ҫ�˽������̼�ļ��������(��֤���������Ƶõ�����ͨ������ʯ��ˮ�����ܻ��ǣ����Ƕ�����̼�������������õ�ȼ��ľ�������ڼ���ƿ�ڣ�ľ��Ϩ��֤���Ѽ���������̼����)����Ҫ���ն�����̼�Ի�����Ӱ��(������̼�����Ӱ�죺�����ŷ���������ЧӦ)�����֪ʶ���Ǵ���Ĺؼ���

 ͨ��ѧ��Ĭд����ϵ�д�
ͨ��ѧ��Ĭд����ϵ�д� ���ƽ̸�������ѡ����ĩ���100��ϵ�д�
���ƽ̸�������ѡ����ĩ���100��ϵ�д�����Ŀ�����ֳ������ʵ�pH��Χ���±�������������ǿ���ǣ� ��
���� | ���� | ������ | ���� | ����ˮ |
pH��Χ�����£� | 4 ~ 5 | 7 ~ 8 | 8 ~ 9 | 10 ~ 11 |
A.����
B.������
C.����
D.����ˮ