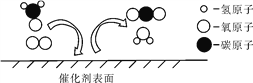
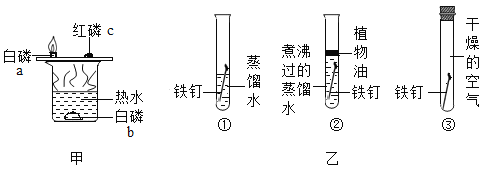
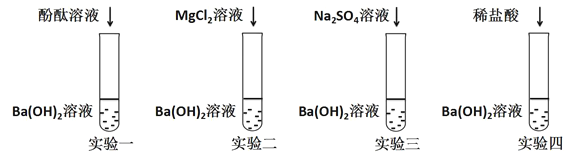
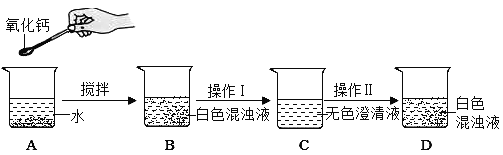
��Ŀ����
������������ʯ�ң�ijͬѧ������������������ʵ�鷽����
��1��A�з�����Ӧ�Ļ�ѧ����ʽΪ_____������B��������IJ������_____
��2�����Ҫ��ʵ��֤��C�в���ˮ����һ����Һ������ѡ������_____������ţ�����ʵ�顣
��ϡ���� �ڷ�̪��Һ �۶�����̼ ���Ȼ�����Һ
��3���������Ǽ���ij������ʹ��ɫ����Һ���ְ�ɫ���壬���������DZ���̼������Һ����ɫ����Ļ�ѧʽ��_____�����������ǹ����������ƣ�ȫ���ܽ⣩����ɫ����Ļ�ѧʽ��_____��
���������ϣ���ҵ������Ҫ�ɷ���Fe2O3��������������FeO��Fe3O4
��1�����ᾧ�壨H2C2O4����3H2O����Ũ�������������ȷֽ⣬��ѧ����ʽ_____��
��2����ʯ���ǹ���NaOH��CaO�Ļ���������ˮ�����Ͷ�����̼��
��3�����ij�������������������������
���������� | FeO | Fe2O3 | Fe3O4 |
������������ | 77.8% | 70.0% | 72.4% |
���������ۣ�Ϊ�˲ⶨ��������������������С��ͬѧ��������ʵ�顣��װ�����������ã�
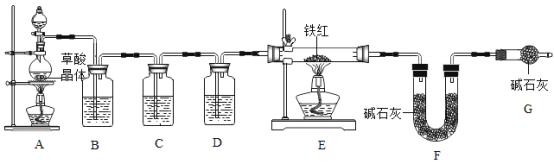
��1����ʵ��Ϊ�˱�֤����E�е������Ǵ����������CO����B��C��D�е��Լ�������_____������ĸ��ţ���
a.Ũ���� b.�����ʯ��ˮ c.����������Һ
��2��Cװ�õ�������_____��
��3��д��Eװ������������Ӧ��һ����ѧ����ʽ��_____��
��4����ȡ������Ʒ10.0g��������װ�ý���ʵ�飬�ⶨ��������������������
����E�г�ַ�Ӧ��õ����۵�����Ϊm g����_____<m��_____��
����ʵ��ǰ��Ƶ�Fװ������7.7g�������������������������_____
��ʵ�鷴˼��
��1�����ȱ��Gװ�ã��������������أ���������Ʒ����������������_____��ѡ�ƫС�������䡱��ƫ����
��2����ʵ��װ�õ�һ������ȱ����_____��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д������ࡱ��ѧϰ��ѧ����Ҫ����֮һ�����з�����ȷ���ǣ�������
ѡ�� ��� | ����� | ���� | ������ | �� |
A | �Ҵ����� | ���ʯ | CaO | ���� |
B | ú | ���� | �ɱ� | H3PO4 |
C | Ӳˮ | �� | Fe2O3 | NH4HCO3 |
D | ���� | He | CH4O | ���� |
A. A B. B C. C D. D