��Ŀ����
��A��B��C��D��֧�Թ��зֱ�ʢ��NaHSO4��Na2CO3��BaCl2��BaCO3���壬ijͬѧ��ֻ������ˮ�������Թܵ������£�������һһ���������
��1��NaHSO4��ˮ�л�����Na+��H+��SO42-�������ʷ���ĽǶȿ��ǣ�NaHSO4���� (A.�� B.�� C.�� D.������)��
��2�����ȼ�������Ĺ��������� ���������ֿ�ֱ�Ӽ�����Ĺ��������� ��
��3��ʣ�����ֹ��屻�������ʱ������Ӧ�Ļ�ѧ����ʽ�ֱ��� �� ��
��1��C ��1�֣�
��2��BaCO3 ��1�֣� NaHSO4 ��1�֣�
��3��2 NaHSO4 + Na2CO3 == 2 Na2SO4 + CO2�� + H2O��2�֣�
BaCl2 + NaHSO4 == BaSO4�� + NaCl + HCl ��2�֣�

��ϰ��ϵ�д�
 ��ְٷְټ�����Ԫ��ĩ���Ծ�ϵ�д�
��ְٷְټ�����Ԫ��ĩ���Ծ�ϵ�д�
�����Ŀ
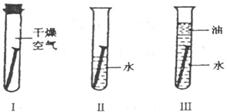
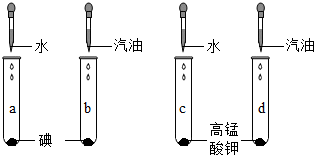 ��A��B��C��D��֧�Թ��и�����1��2С����������أ�Ȼ��ֱ����5mLˮ�����ͣ���ͼ��ʾ�������۲�����
��A��B��C��D��֧�Թ��и�����1��2С����������أ�Ȼ��ֱ����5mLˮ�����ͣ���ͼ��ʾ�������۲�����