��Ŀ����
����Ŀ��ʵ������ȡ���峣���õ�����װ�ã����ݸ�����װ�ûش��������⣺ 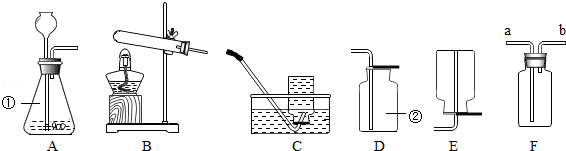
��1��д��������������ƣ������� ��
��2������ʵ��ѡ��װ��A����ȡ��������д���÷�Ӧ�Ļ�ѧ����ʽ ��
��3����ѡ����������ȡ���ռ��ϴ�����O2 �� ��Ӧѡ��װ�� �� ����װ����һ������ĵط�������ָ�������ԸĽ��� ��
��4����ͼFװ�ÿ����ռ����������������ƿ����ˮ���ռ�����������Ӧ�����a����b������ͨ�룻�������a��ͨ����ƿ�����ƿ��Ӧʢװ�����Լ������ƣ�������������
��5��ijͬѧ������װ��ͼ��ѡ���ʵ�װ�óɹ����Ʊ����ռ��˶�����̼��ѡ�õ�װ����������ĸ�����÷�Ӧ�Ļ�ѧ����ʽ�� �� Ϊ�˽�һ����֤�����������Ƕ�����̼����ͬѧ������ͨ��ͼFװ���У���Fװ����Ӧ������Լ�Ϊ���ѧʽ��������Fװ���м�����ɫʯ����Һ���۲쵽�������� �� �ó��Ľ���
���𰸡�
��1����ƿ������ƿ
��2��2H2O2 ![]() 2H2O+O2��
2H2O+O2��
��3��B���Թ��ڼ�һ����
��4��b��Ũ����
��5��AD��CaCO3+2HCl=CaCl2+H2O+CO2����Ca��OH��2����죻CO2����ˮ����Һ������
���������⣺��1����������������Ҫ��д��ȷ����2������Aװ����ȡ����������װ��û�оƾ��ƣ�����Ӧ�ù���������ȡ��������3�����������ȡ������Ҫע��ʹ����������֤������������ط۳������ԸĽ���ʹ�ö�Ҫע��õ㣮��4��������ռ�������ǰһװ��Ϊ�ռ�װ�ã�����Ӧ���Ƕ̽�����������һװ��Ϊ����װ�ã�����Ӧ���dz����̳���ͬʱѡ��Ũ���������������5��������̼���ʵļ��飬��������ɫʯ��ʱʱҪ��֤��Ӧ��Һ������ԣ���Ҫ������ָʾ���ı�ɫ����� ���Դ��ǣ���1������ƿ�ڼ���ƿ��2��2H2O2 ![]() 2H2O+O2����3��BC����ѡ����ѡ��©ѡ�����÷֣�
2H2O+O2����3��BC����ѡ����ѡ��©ѡ�����÷֣�
���Թܿ�Ӧ��һ��������ֹKMnO4�Ŀ������������뵼���ܣ����������ܣ��������𰸾��ɵ÷֣� ��4��b ŨH2SO4��5��AD����ѡ����ѡ��©ѡ�����÷֣� CaCO3+2HCl=CaCl2+H2O+CO2��Ca��OH��2
��Һ���CO2����ˮ����Һ�����ԣ���CO2�����Բ��÷֣�

����Ŀ��Ϊ�ⶨijBaCl2��Һ������������������������ʵ�飺ȡ104g����Һ���ձ��У�Ȼ��30gһ����������������������Һ�����μ��룬ÿ�γ�ַ�Ӧ����ˡ�������ʵ�������ʵ�������ʧ���Բ��ƣ������������
��1�� | ��2�� | ��3�� | |
����������Һ������/g | 10 | 10 | 10 |
���˺���Һ������/g | 104 | 104 | 110.7 |
�����������ݼ��㣺��BaCl2+H2SO4�TBaSO4��+2HCl��
��1����ʵ������в���������������Ϊg��
��2����BaCl2��Һ�����ʵ�����������д��������̣���