��Ŀ����
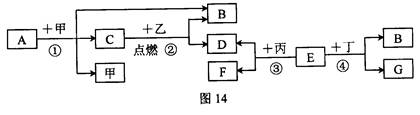
��2009��̩�ݡ�A�dz����������ʣ�B��C�Ƿǽ������ʣ���C���ܶ���С�����壬���Ǵ��������Ҫ�ɷ֣�������ɫҺ�壬����֮�������µ�ת����ϵ(���ֲ���ͷ�Ӧ����δ����)��
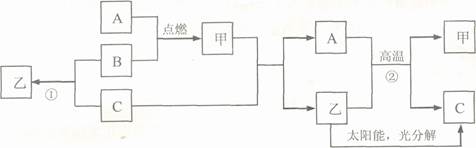
(1)�ƶ�C���ס��ҵĻ�ѧʽΪ��C ���� ���� ��
(2)д����Ӧ�ڵĻ�ѧ����ʽ ��
(3)д����Ӧ�ٵĻ�ѧ����ʽ ���÷�Ӧ���˾��зų������࣬ԭ�ϲ������Ƶ��ŵ��⣬���� ���ŵ㡣

(1)�ƶ�C���ס��ҵĻ�ѧʽΪ��C ���� ���� ��
(2)д����Ӧ�ڵĻ�ѧ����ʽ ��
(3)д����Ӧ�ٵĻ�ѧ����ʽ ���÷�Ӧ���˾��зų������࣬ԭ�ϲ������Ƶ��ŵ��⣬���� ���ŵ㡣
(1) H2 Fe3O4 H2O
(2) 3Fe��4H2O ="===" Fe3O4��4H2��
(3) 2H2��O2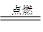 2H2O ��������Ⱦ
2H2O ��������Ⱦ
(2) 3Fe��4H2O ="===" Fe3O4��4H2��
(3) 2H2��O2
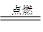 2H2O ��������Ⱦ
2H2O ��������Ⱦ��1�����������Ҫ�ɷ�������������������������ȼ��������������������A��B�������ʷֱ���������������������������ѧʽ�ֱ�Ϊ��H2��Fe3O4��H2O�����H2��Fe3O4��H2O��
��2������ˮ�ڸ��������·�Ӧ��������������������������Ӧ�Ļ�ѧ����ʽΪ��3Fe+4H2O==Fe3O4+4H2��
��3������ȼ��������ˮ����Ӧ�Ļ�ѧ����ʽΪ��2H2+O2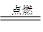 2H2O��
2H2O��
�÷�Ӧ���˾��зų������࣬ԭ�ϲ������Ƶ��ŵ��⣬��������Ⱦ�������������Ⱦ��
��2������ˮ�ڸ��������·�Ӧ��������������������������Ӧ�Ļ�ѧ����ʽΪ��3Fe+4H2O==Fe3O4+4H2��
��3������ȼ��������ˮ����Ӧ�Ļ�ѧ����ʽΪ��2H2+O2
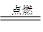 2H2O��
2H2O���÷�Ӧ���˾��зų������࣬ԭ�ϲ������Ƶ��ŵ��⣬��������Ⱦ�������������Ⱦ��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ