��Ŀ����
����Ŀ��ij��ѧ��ȤС��ͬѧΪ̽������Ļ�ѧ���ʣ���������ʵ�飺

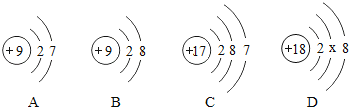
��1���Թܢ��е�������_____����Ӧ���Թܢ���һ���е���������_____��д���ӷ��ţ���
��2���Թܢ��з�Ӧ�Ļ�ѧ����ʽΪ_____��
��3��ʵ���з����Թܢ��й���ȫ���ܽ⣬�Թܢ��в�����ɫ�������������Թܢ��еμ�����������Һ�������������������������������Һ��������ϵ����ͼ2��ʾ�����Թܢ��е�������_____��ͼ2��a��b�ζ�Ӧ��Һ��һ��������Ӧ�Ļ�ѧ����ʽΪ_____��
���𰸡� ��� Fe2+ Fe2O3+3H2SO4==Fe2(SO4)3+3H2O H2SO4��Fe2��SO4��3 2NaOH+H2SO4�TNa2SO4+2H2O
����������1����ʯ����Һ��ϡ�����졣��Fe +2HCl== FeCl2+H2���� FeCl2������ˮ���ʷ�Ӧ���Թܢ���һ���е��������� Fe2+����2���Թܢ��з�Ӧ�Ļ�ѧ����ʽFe2O3+3H2SO4==Fe2(SO4)3+3H2O��3��ʵ���з����Թܢ��й���ȫ���ܽ⣬�Թܢ��в�����ɫ�������������Թܢ��еμ�����������Һ������ͼ���֪����ʼ��������������Һʱ����������������˵����ϡ����ʣ�࣬�Թܢ��е�������ʣ���H2SO4�����ɵ�Fe2(SO4)3��ͼ2��a��b����ϡ�������������Ʒ�Ӧ����ѧ����ʽΪ��2NaOH+H2SO4===Na2SO4+2H2O

 ��ѧ����ͬ����ϰϵ�д�
��ѧ����ͬ����ϰϵ�д� ��ǰ�κ�ͬ����ϰϵ�д�
��ǰ�κ�ͬ����ϰϵ�д� ����С��ҵϵ�д�
����С��ҵϵ�д� �Ƹ�С״Ԫ����������ϰ��ϵ�д�
�Ƹ�С״Ԫ����������ϰ��ϵ�д� �ɹ�ѵ���ƻ�ϵ�д�
�ɹ�ѵ���ƻ�ϵ�д� ����ѵ����ֱͨ�п�����ϵ�д�
����ѵ����ֱͨ�п�����ϵ�д�