��Ŀ����
��ѧϰ����кͷ�Ӧʱ��ͬѧ������ϡ���������������Һ��Ӧ��ʵ�顣
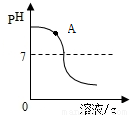
��1��������ʵ������вⶨ������һ����Һ��pH�仯����ͼ��֪����С��ͬѧ�ⶨ����___________��Һ��pH�仯���÷�Ӧ�Ļ�ѧ����ʽΪ___________��ʵ������У�ͬѧ��ȡ����ͼ��A��������Һ�������Լ�_____________ ������ţ����۲쵽����ɫ�������ɡ�
��BaCl2��Һ
��Fe2(SO4)3��Һ
��CuSO4��Һ���ۣ����û��ǡ���кͣ�A����Һ�е�����Ϊ____________��
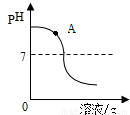
��1��������ʵ������вⶨ������һ����Һ��pH�仯����ͼ��֪����С��ͬѧ�ⶨ����___________��Һ��pH�仯���÷�Ӧ�Ļ�ѧ����ʽΪ___________��ʵ������У�ͬѧ��ȡ����ͼ��A��������Һ�������Լ�_____________ ������ţ����۲쵽����ɫ�������ɡ�
��BaCl2��Һ
��Fe2(SO4)3��Һ
��CuSO4��Һ���ۣ����û��ǡ���кͣ�A����Һ�е�����Ϊ____________��
��2����������з�̪������������Һ�еμ�ϡ���ᣬ�ɹ۲쵽____________ �����ۣ�������ǡ���кͣ�Ҳ�������������Ϊ�˽�һ��ȷ����Ӧ��������Һ���Ƿ������ᣬ����ͬѧ������������ַ���������һ��ȡ��Ӧ����Һ���Թ��У��μ�̼������Һ�����۲쵽___________���������������������ȡ��Ӧ����Һ���Թ��У��μ�����������Һ����������ɫ������ ���������������Ϊ�������ַ��������ֲ���������˵������_____________��
��1��NaOH ��2NaOH +H2SO4��Na2SO4+2H2O����Na2SO4 ��NaOH
��2����ɫ��Ϊ��ɫ��ð���ݣ���������Na2SO4��Ba(OH)2��ӦҲ���ɰ�ɫ����
��2����ɫ��Ϊ��ɫ��ð���ݣ���������Na2SO4��Ba(OH)2��ӦҲ���ɰ�ɫ����

��ϰ��ϵ�д�
 �������¿��ÿ�ʱ��ҵϵ�д�
�������¿��ÿ�ʱ��ҵϵ�д� Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
�����Ŀ
 ��2012?�ֶ�������ģ����ѧϰ����кͷ�Ӧʱ��ͬѧ������ϡ���������������Һ��Ӧ��ʵ�飮
��2012?�ֶ�������ģ����ѧϰ����кͷ�Ӧʱ��ͬѧ������ϡ���������������Һ��Ӧ��ʵ�飮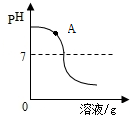 ��ѧϰ����кͷ�Ӧʱ��ͬѧ������ϡ���������������Һ��Ӧ��ʵ�飮
��ѧϰ����кͷ�Ӧʱ��ͬѧ������ϡ���������������Һ��Ӧ��ʵ�飮