��Ŀ����
ijѧ��Ϊ�ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ������������������ͼ��ʾ��װ�ã�ͼ������̨����ȥ����ʵ����
27�棬l01kPa�½��У�ʵ�鲽�����£�
�ٰ�ͼ���Ӻ�װ�ã�
��______��
������ƽȷ��ȡ����������Ʒ6g������A���Թ��ڣ���B�м���ƿ��װ�����Ͷ�����̼ˮ��Һ��
�����Һ©���м���ϡ���ᣬ��������ϡ��������Թ������������رջ�������Ӧ��������Ͳ���ռ������Ͷ�����̼ˮ��Һ220mL��
��ش��������⣺
��1����ʵ�鲽��١���֮�䣬��ȱ��һ�����裬�뽫�䲹���ڢں���ĺ����ϣ�
��2���ж��������Ʒ������ʵ�ʵ��������______���������Ʊ��ʵ�ԭ����______ ���û�ѧ����ʽ��ʾ����
��3��B�м���ƿʢװ���DZ��Ͷ�����̼ˮ��Һ������ˮ����������______��
��4���жϲ�����е��˵�ϡ�����ѹ����ı�־��______��
��5������������Ʒ��̼���Ƶ���������Ϊ______��27�棬l01kPa�¶�����̼���ܶ�Ϊl.8g/L��������̼����������ų��ı��Ͷ�����̼ˮ��Һ���������
��6��Ҫ��ȥ���ֱ������������е�̼���ƣ�Ӧ����������������Ʒ����ˮ�����Һ��Ȼ������Һ�м�������______����ַ�Ӧ����ˡ�������Һ�����ܵõ��Ƚϴ������������ƹ��壮��Ӧ����ʽΪ______��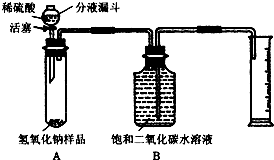
27�棬l01kPa�½��У�ʵ�鲽�����£�
�ٰ�ͼ���Ӻ�װ�ã�
��______��
������ƽȷ��ȡ����������Ʒ6g������A���Թ��ڣ���B�м���ƿ��װ�����Ͷ�����̼ˮ��Һ��
�����Һ©���м���ϡ���ᣬ��������ϡ��������Թ������������رջ�������Ӧ��������Ͳ���ռ������Ͷ�����̼ˮ��Һ220mL��
��ش��������⣺
��1����ʵ�鲽��١���֮�䣬��ȱ��һ�����裬�뽫�䲹���ڢں���ĺ����ϣ�
��2���ж��������Ʒ������ʵ�ʵ��������______���������Ʊ��ʵ�ԭ����______ ���û�ѧ����ʽ��ʾ����
��3��B�м���ƿʢװ���DZ��Ͷ�����̼ˮ��Һ������ˮ����������______��
��4���жϲ�����е��˵�ϡ�����ѹ����ı�־��______��
��5������������Ʒ��̼���Ƶ���������Ϊ______��27�棬l01kPa�¶�����̼���ܶ�Ϊl.8g/L��������̼����������ų��ı��Ͷ�����̼ˮ��Һ���������
��6��Ҫ��ȥ���ֱ������������е�̼���ƣ�Ӧ����������������Ʒ����ˮ�����Һ��Ȼ������Һ�м�������______����ַ�Ӧ����ˡ�������Һ�����ܵõ��Ƚϴ������������ƹ��壮��Ӧ����ʽΪ______��

��1����װ��Ҫ�����������ã�����ȷ������������̼�����������ʵ��ǰӦ�ȼ��װ�õ������ԣ�
��2�����������Ǽ���ԺͿ����еĶ�����̼��Ӧ������̼���ƶ����ʣ����������ɵ�̼������ϡ��������ɶ�����̼���壮
��3�������ʵ��Ŀ����ͨ���������̼�����������ҩƷ��̼���Ƶĺ��������Ա��뱣֤������̼��ȷ�ԣ���Ϊ������̼��������ˮ�������ñ��͵Ķ�����̼ˮ��Һ�����Ա��������̼�ļ��٣�
��4��֤�������������֤��̼���Ʒ�Ӧ�꣬���Կ��Լ�����һ�����ᣬû�����ݲ�����
��5�����ɶ�����̼������Ϊ��
L��1.8 g/L=0.396 g
����Ʒ�к�̼���Ƶ�����Ϊx
Na2CO3+H2SO4=Na2SO4+CO2��+H2O
106 44
x 0.396g
=
��
��֮�ã�x=0.954g
����Ʒ��̼���Ƶ���������=
��100%=15.9%
��6������������Һ���ʺ����̼���ƣ��������ƿ��Ժ�̼���Ʒ�����Ӧ������̼��Ƴ�������Ҫ�������ֱ��ʵ�����������Һ�е����ʣ����Լ���Ba��OH��2��Һ�����˳���Ӧ���ɵ�BaCO3��
�ʴ�Ϊ����1�����װ��������
��2��������������Ʒ�еμ�ϡ����ʱ�������� 2NaOH+CO2�TNa2CO3+H2O
��3��CO2������ˮ���������ڱ��͵�CO2ˮ��Һ��
��4��װ��A���Թ���û�����ݲ���
��5��15.9%
��6��Ba��OH��2��Ca��OH��2 Ba��OH��2+Na2CO3�TBaCO3��+2NaOH��Ca��OH��2+Na2CO3�TCaCO3��+2NaOH
��2�����������Ǽ���ԺͿ����еĶ�����̼��Ӧ������̼���ƶ����ʣ����������ɵ�̼������ϡ��������ɶ�����̼���壮
��3�������ʵ��Ŀ����ͨ���������̼�����������ҩƷ��̼���Ƶĺ��������Ա��뱣֤������̼��ȷ�ԣ���Ϊ������̼��������ˮ�������ñ��͵Ķ�����̼ˮ��Һ�����Ա��������̼�ļ��٣�
��4��֤�������������֤��̼���Ʒ�Ӧ�꣬���Կ��Լ�����һ�����ᣬû�����ݲ�����
��5�����ɶ�����̼������Ϊ��
| 220 |
| 1000 |
����Ʒ�к�̼���Ƶ�����Ϊx
Na2CO3+H2SO4=Na2SO4+CO2��+H2O
106 44
x 0.396g
| 106 |
| x |
| 44 |
| 0.396g |
��֮�ã�x=0.954g
����Ʒ��̼���Ƶ���������=
| 0.954g |
| 6g |
��6������������Һ���ʺ����̼���ƣ��������ƿ��Ժ�̼���Ʒ�����Ӧ������̼��Ƴ�������Ҫ�������ֱ��ʵ�����������Һ�е����ʣ����Լ���Ba��OH��2��Һ�����˳���Ӧ���ɵ�BaCO3��
�ʴ�Ϊ����1�����װ��������
��2��������������Ʒ�еμ�ϡ����ʱ�������� 2NaOH+CO2�TNa2CO3+H2O
��3��CO2������ˮ���������ڱ��͵�CO2ˮ��Һ��
��4��װ��A���Թ���û�����ݲ���
��5��15.9%
��6��Ba��OH��2��Ca��OH��2 Ba��OH��2+Na2CO3�TBaCO3��+2NaOH��Ca��OH��2+Na2CO3�TCaCO3��+2NaOH

��ϰ��ϵ�д�
�����Ŀ
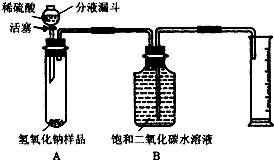 ��2009?��Ԫ��ijѧ��Ϊ�ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ������������������ͼ��ʾ��װ�ã�ͼ������̨����ȥ����ʵ����
��2009?��Ԫ��ijѧ��Ϊ�ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ������������������ͼ��ʾ��װ�ã�ͼ������̨����ȥ����ʵ����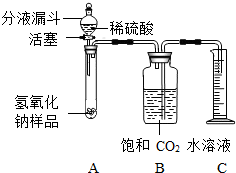 ijѧ��Ϊ�˲ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ��������������������ͼ��ʾ��װ�ã�ͼ������̨����ȥ����ʵ����27�棬101kPa�½��У�ʵ�鲽�����£�
ijѧ��Ϊ�˲ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ��������������������ͼ��ʾ��װ�ã�ͼ������̨����ȥ����ʵ����27�棬101kPa�½��У�ʵ�鲽�����£� ijѧ��Ϊ�ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ������������������ͼ��ʾ��װ�ã�ͼ������̨����ȥ����ʵ����
ijѧ��Ϊ�ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ������������������ͼ��ʾ��װ�ã�ͼ������̨����ȥ����ʵ����