��Ŀ����
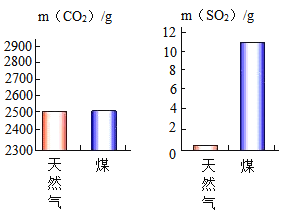
����Ŀ����ͼ�ǹ�ҵ�����Ȼ������������ƺ����������ͼ���ش��������⡣

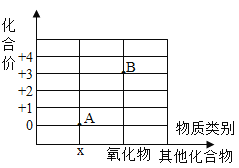
��1���ڳ����£�һ������ˮ��������Ʊ���ʳ��ˮ_____ ��
��2�����������׳�_____������������¶�ڿ��������ױ��ʣ�д�������ʵĻ�ѧ����ʽ_____��
��3���ڷ�Ӧ����ͨ��ʱ��������������_____���� ����������������������д����Ӧ�ڵĻ�ѧ����ʽ_____��
��4��Ũ������¶�ڿ����г��ֵ�������_____����ҵ���᳣�ʻ�ɫ������Ϊ���к������ʣ�����Ϊ���ܺ��е�������_____������ ���Ȼ������������Ȼ�ͭ�������Ȼ�������
���𰸡���ˮ�м���ʳ�ν���ֱ������Ϊֹ ���ռ������� CO2+2NaOH==Na2CO3+H2O ���� Cl2+H2![]() 2HCl �������� �Ȼ���
2HCl �������� �Ȼ���
��������
��1���ڳ����£�һ������ˮ�����Ʊ���ʳ��ˮ�ķ����ǣ���ˮ�м���ʳ�ν���ֱ������Ϊֹ��
��2�����������׳ƻ��ռ������ƣ��ڿ����У����������������̼��Ӧ����̼���ƺ�ˮ������������¶�ڿ��������ױ��ʣ������ʵĻ�ѧ����ʽΪ��CO2+2NaOH==Na2CO3+H2O��
��3��ˮͨ��ʱ�����������������������������������Ϊ2��1���ڷ�Ӧ����ͨ��ʱ�������������ڸ�������Ӧ����������������ȼ�������Ȼ��⣬��Ӧ�Ļ�ѧ����ʽΪ��Cl2+H2![]() 2HCl��
2HCl��
��4��Ũ������лӷ��ԣ��ӷ������Ȼ������ڿ����е�ˮ�������γ������СҺ�Ρ�Ũ������¶�ڿ����г��ֵ������Dz������������������ӵ���Һ�ʻ�ɫ����ҵ���᳣�ʻ�ɫ������Ϊ���к������ʣ����ܺ��е��������Ȼ�����