��Ŀ����
�������仯�����������������й㷺��Ӧ�á�
��һ��������Ӧ�úͷ���
��1���ɻ���ȼ���Ǻ���ú�ͣ�����Ҫ����____���Ƶģ��ʯ�͡���ú������
��2����������Ҫͬʱ�Ӵ�O2��____�����������������Ʒ�����һ�ַ���____��
��3�����ָ��е�Mn������MnO2��Al�ۻ���ڸ�����ͨ���û���Ӧ�Ƶã��û�ѧ����ʽΪ____��
��4��Mg��OH��2���ȷֽ�����MgO��Mg��OH��2������ȼ������ԭ����____������ĸ����
A �ֽ����ȣ����ͻ����¶� B MgO����ȼ�����������
�������Ʊ���ԭ������
ʵ�������÷���м��������Fe2O3��Ϊԭ���Ʊ���ԭ�����۵��������£�

��1��Fe�ۿ�ʹ�����ܡ�������Fe2��SO4��3ת��ΪFeSO4���÷�Ӧ�Ļ�ѧ����ʽΪ____��
��2������FeCO3��ϴ���ķ�����ȡ���һ��ϴ�Ӻ����Һ���ȼ���һ����ϡ���ᣬ��ȥCO32-���ٵμ�____��Һ������������
��3�����������ת����FeCO3 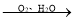 FeOOH+CO2����ת���Ļ�ѧ����ʽΪ____��
FeOOH+CO2����ת���Ļ�ѧ����ʽΪ____��
��4�������ա�ǰ��Ӧ��̿���롰������ù�����Ȼ�ϣ�Ŀ����____��
�������ⶨ��ԭ�����۵����
��ԭ�������к�������FexCy��С��ͬѧ����ʦ��ָ���½�������ʵ�顣
���ϣ��� Fe��FexCy�ڼ���ʱ��O2��Ӧ������Fe2O3��CO2��
��FexCy�����ᷴӦ��
ʵ��1��ȡ30.48g��Ʒ����������ϡH2SO4����ַ�Ӧ������1.00gH2��
ʵ��2����ȡ30.48g��Ʒ������ͼ����ʵ�顣

����װ��A��B�������仯
װ�� | A | B |
��Ӧǰ | 400.00g | 400.00g |
��Ӧ�� | mg | 400.88g |
ʵ���A�й���ȫ����Ϊ����ɫ��װ��A��B�������仯���ϱ���
��1��װ���װ�ú�ʵ��ǰ��Ҫ____��
��2��װ��C��������____��
��3��FexCy�У�x��y=____������m��ֵ��____g��
��ʵ�鷴˼�������ÿ�������ȥCO2�������O2����ʵ�飬���x��y��ֵƫ���ܵ�ԭ����____��
 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�


 ��ͭ���Ȼ�����Һ�з�����Ӧ��
��ͭ���Ȼ�����Һ�з�����Ӧ��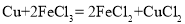 �������Ȼ�����Һ�з�����Ӧ��
�������Ȼ�����Һ�з�����Ӧ��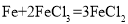 ��
��