��Ŀ����
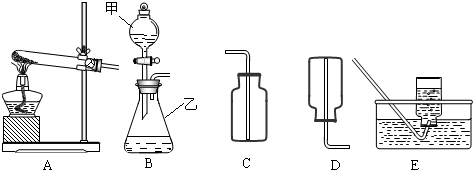
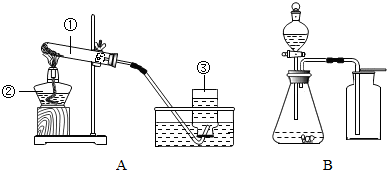 ʵ���ҳ�����ͼ��ʾװ����ȡ�й����壮
ʵ���ҳ�����ͼ��ʾװ����ȡ�й����壮��ش��������⣺
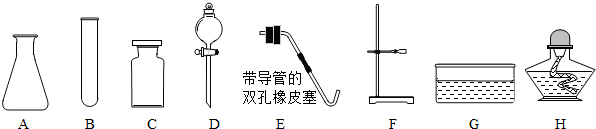
��1��д��������������ƣ�
��
�Թ�
�Թ�
�����ƾ���
�ƾ���
��������ƿ
����ƿ
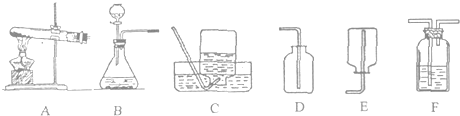
����2����KMnO4��������ȡO2��Ӧѡ����ͼ�е�
A
A
װ�ã���A��B������3��ʵ������ϡ����ʹ���ʯ��ȡCO2ѡ����ͼ�е�
B
B
װ�ã���A��B�����÷�Ӧ�Ļ�ѧ����ʽΪCaCO3+2HCl�TCaCl2+H2O+CO2��
CaCO3+2HCl�TCaCl2+H2O+CO2��
�����ô�װ��Ҳ����ȡ��������Ӧ�Ļ�ѧ����ʽΪ��2H2O2
2H2O+O2��
| ||
2H2O2
2H2O+O2��
��
| ||
��4��ʵ������ȡ���⣨H2S�����壬һ����FeS�����壩�����ᷴӦ�Ƶã���֪H2S����������ˮ���ܶȱȿ���������Ҫ��ȡH2S��Ӧѡ����ͼ��
B
B
����A��B����ͼ�е�H2S�ռ���������������
��������
������������1��ֱ��д�����������ƣ�
��2����KMnO4��������ȡ�������ڹ̹̼����͵ķ�Ӧ�����Կ��Ծݴ�ѡ����ȡװ�ã�
��3��ʵ������ϡ����ʹ���ʯ��ȡCO2���ڹ�Һ�����͵ķ�Ӧ��������ȣ�����ϡ����ʹ���ʯ����Ҫ�ɷ�̼��Ʒ�Ӧ�������Ȼ��ơ�������̼��ˮ����װ��Ҳ�����ù���������Һ����ȡ���������й�������ֽ������ˮ��������
��4������ʵ��������ȡ����ķ�Ӧԭ����������ܶ������н��
��2����KMnO4��������ȡ�������ڹ̹̼����͵ķ�Ӧ�����Կ��Ծݴ�ѡ����ȡװ�ã�
��3��ʵ������ϡ����ʹ���ʯ��ȡCO2���ڹ�Һ�����͵ķ�Ӧ��������ȣ�����ϡ����ʹ���ʯ����Ҫ�ɷ�̼��Ʒ�Ӧ�������Ȼ��ơ�������̼��ˮ����װ��Ҳ�����ù���������Һ����ȡ���������й�������ֽ������ˮ��������
��4������ʵ��������ȡ����ķ�Ӧԭ����������ܶ������н��
����⣺��1��ֱ��д������������Ϊ�����Թܣ��ھƾ��ƣ��ۼ���ƿ��
��2����KMnO4������ȡ�������ڹ̹̼����͵ķ�Ӧ������Ӧ��ѡ��A��Ϊ��ȡװ�ã�
��3��ʵ������ϡ����ʹ���ʯ��ȡCO2���ڹ�Һ�����͵ķ�Ӧ��������ȣ�����Ӧ��ѡBװ����Ϊ��ȡװ�ã�����ϡ����ʹ���ʯ����Ҫ�ɷ�̼��Ʒ�Ӧ�������Ȼ��ơ�������̼��ˮ���÷�Ӧ�Ļ�ѧ����ʽΪ��CaCO3+2HCl=CaCl2+H2O+CO2������װ��Ҳ�����ù���������Һ����ȡ���������й�������ֽ������ˮ���������÷�Ӧ�Ļ�ѧ����ʽΪ��2H2O2
2H2O+O2����
��4������ʵ��������ȡ����ķ�Ӧԭ������֪������ȡ���������ҩƷ��״̬Ϊ�����Һ�壬������Ϊ���£�������ܶȵ��ܶȱȿ�������������ˮ������Ӧ��ѡ���Һ�����͵ķ���װ�ã����������ſ��������ռ�����Bװ������ȡ�������壮
�ʴ�Ϊ����1���Թܣ��ƾ��ƣ�����ƿ��
��2��A��
��3��B��CaCO3+2HCl�TCaCl2+H2O+CO2����2H2O2
2H2O+O2����
��4��B������������
��2����KMnO4������ȡ�������ڹ̹̼����͵ķ�Ӧ������Ӧ��ѡ��A��Ϊ��ȡװ�ã�
��3��ʵ������ϡ����ʹ���ʯ��ȡCO2���ڹ�Һ�����͵ķ�Ӧ��������ȣ�����Ӧ��ѡBװ����Ϊ��ȡװ�ã�����ϡ����ʹ���ʯ����Ҫ�ɷ�̼��Ʒ�Ӧ�������Ȼ��ơ�������̼��ˮ���÷�Ӧ�Ļ�ѧ����ʽΪ��CaCO3+2HCl=CaCl2+H2O+CO2������װ��Ҳ�����ù���������Һ����ȡ���������й�������ֽ������ˮ���������÷�Ӧ�Ļ�ѧ����ʽΪ��2H2O2
| ||
��4������ʵ��������ȡ����ķ�Ӧԭ������֪������ȡ���������ҩƷ��״̬Ϊ�����Һ�壬������Ϊ���£�������ܶȵ��ܶȱȿ�������������ˮ������Ӧ��ѡ���Һ�����͵ķ���װ�ã����������ſ��������ռ�����Bװ������ȡ�������壮
�ʴ�Ϊ����1���Թܣ��ƾ��ƣ�����ƿ��
��2��A��
��3��B��CaCO3+2HCl�TCaCl2+H2O+CO2����2H2O2
| ||
��4��B������������
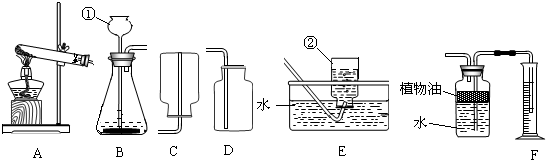
������������Ҫ�������������ȡ��Ҫ�����������Ŀ����Ҫ��dz�������ķ���װ�ú��ռ�װ����ѡȡ�����������ݣ�����������������̼��ʵ������ȡ��������ʱ����Ū��ͼʾ��Ϣ�ȵȣ�

��ϰ��ϵ�д�
�����Ŀ
 ͨ����ѧ�γ̵�ѧϰ�������ջ�࣮ܶ
ͨ����ѧ�γ̵�ѧϰ�������ջ�࣮ܶ